carbohydrate → ugljikohidrat
Carbohydrates (often called carbs for short) are polyhydroxy aldehydes or ketones, or substances that yield such compounds on hydrolysis. They are also known as saccharides, a term derived from the Latin word saccharum for sugar. Carbohydrates are the most abundant class of compounds in the biological world, making up more than 50 % of the dry weight of the Earth’s biomass. Every type of food we eat can have its energy traced back to a plant. Plants use carbon dioxide and water to make glucose, a simple sugar, in photosynthesis. Other carbohydrates such as cellulose and starch are made from the glucose. Light from the sun is absorbed by chlorophyll and this is converted to the energy necessary to biosynthesize carbohydrates
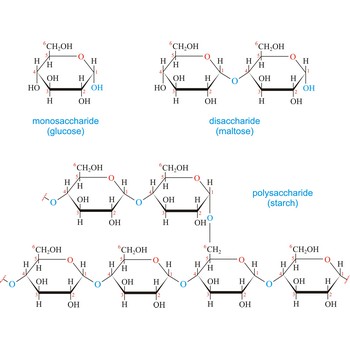
The term carbohydrate was applied originally to monosaccharides, in recognition of the fact that their empirical composition can be expressed as Cx(H2O)y. Later structural studies revealed that these compounds were not hydrates but the term carbohydrate persists.
Carbohydrates are generally classed as either simple or complex. Simple sugars, or monosaccharides, are carbohydrates that can’t be converted into smaller subunits by hydrolysis. Complex carbohydrates are made of two (disaccharides) or more (oligosaccharides, polysaccharides) simple sugars linked together by acetal (glycosidic) bonds and can be split into the former by hydrolysis.
decomposition → raspadanje
Decomposition occurs when chemical compounds are broken up into simple molecules, and even as far as their original elements. These processes are normally irreversible. An example of decomposition is when ammonium nitrate is heated. This produces nitrous oxide and water which are unable to recombine.
harmonic motion → harmoničko gibanje
Harmonic motion is caused by restoring force, acting on a body that is displaced from its equilibrium position. This force tries to put the body back in equilibrium. Usual examples are the motion of a body attached to elastic spring (see: Hooke’s law) and the motion of mathematical pendulum. The body undergoes periodic motion around the equilibrium point.
high-density polyethylene → polietilen visoke gustoće
High-density polyethylene (HDPE) is a thermoplastic polyolefin with a density of from 0.941 g/cm3 to 0.960 g/cm3. It is a recyclable plastic, used for items such as milk containers, detergent containers, and base cups of plastic soft drink bottles. HDPE has a high degree of resistance to chemicals, is easy to keep clean, and easily welded.
inorganic chemistry → anorganska kemija
Inorganic chemistry, the branch of chemistry concerned with compounds of elements other than carbon. Certain simple carbon compounds, such as CO, CO2, CS2, carbonates CO32- and cyanides CN-, are usually treated in inorganic chemistry.
dioxin → dioksin
Dioxin is a general term that describes a group of hundreds of chemicals that are highly persistent in the environment. The most toxic compound is 2,3,7,8-tetrachlorodibenzo-p-dioxin or TCDD. The toxicity of other dioxins and chemicals like PCBs that act like dioxin are measured in relation to TCDD. Dioxin is formed as an unintentional by-product of many industrial processes involving chlorine such as waste incineration, chemical and pesticide manufacturing and pulp and paper bleaching. Dioxin was the primary toxic component of Agent Orange, found at Love Canal in Niagara Falls, NY and was the basis for evacuations at Times Beach, MO and Seveso, Italy.
Dioxin is formed by burning chlorine-based chemical compounds with hydrocarbons. The major source of dioxin in the environment comes from waste-burning incinerators of various sorts and also from backyard burn-barrels. Dioxin pollution is also affiliated with paper mills which use chlorine bleaching in their process, with the production of Polyvinyl Chloride (PVC) plastics, and with the production of certain chlorinated chemicals (like many pesticides).
dissociation → disocijacija
Dissociation is the process by which a chemical combination breaks up into simpler constituents as a result of either added energy (dissociated by heat), or the effect of a solvent on a dissolved polar compound (electrolytic dissociation). It may occur in the gaseous, solid, or liquid state, or in a solution.
An example of dissociation is the reversible reaction of hydrogen iodide at high temperatures
The term dissociation is also applied to ionisation reactions of acids and bases in water. For example
which is often regarded as a straightforward dissociation into ions
invertase → invertaza
Invertase (sucrase, saccharase, beta-fructofuranosidase) is an enzyme present in yeast and in the intestinal juice of animals that catalyze the hydrolysis of table sugar (sucrose, saccharose) to the simple sugars, glucose and fructose. This equimolar mixture of glucose and fructose is called invert sugar.
mineralization → mineralizacija
Mineralization is a process in which organic compounds (e.g. dead plant or animal material) are converted to inorganic compounds (e.g. nitrate, carbon dioxide).
Citing this page:
Generalic, Eni. "Jednostavna sol." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table