alkalinity → alkalitet
Alkalinity is a measure of a material’s ability to neutralize acids. It is usually determined using titration.
litmus paper → lakmus-papir
Litmus paper is an unsized paper treated with litmus for the use as an acid-base indicator, in an acid it turns red, and in a base it turns blue.
EDTA → EDTA
Ethyldiaminetetraacetic acid (C10H16N2O8) or shortened EDTA is a hexadentant ligand, and it forms chelates with both transition-metal ions and main-group ions. EDTA is used as a negative ion - EDTA4-. The diagram shows the structure of the ion with the important atoms picked out. The EDTA ion entirely wraps up a metal ion using all 6 of the positions. The co-ordination number is again 6 because of the 6 co-ordinate bonds being formed by the central metal ion.
EDTA is frequently used in soaps and detergents, because it forms a complexes with calcium and magnesium ions. These ions are in hard water and interfere with the cleaning action of soaps and detergents. EDTA is also used extensively as a stabilizing agent in the food industry and as an anticoagulant for stored blood in blood banks. EDTA is the most common reagent in complexometric titration.
Zimmermann-Reinhardt’s reagent → Zimmermann-Reinhardtov reagens
Zimmermann-Reinhardt’s reagent is a mixture of manganese(II) sulphate, sulphuric acid and phosphorus acid. It is used for preventing oxidation of chloride ion while titrating iron(II) ion with permanganate solution.
histidine → histidin
Histidine is an electrically charged amino acids with basic side chains. It is an essential amino acid, which means that humans cannot synthesize it, so it must be ingested. Histidine is perhaps the most common and versatile catalytic residue in proteins. The imidazole sidechain of histidine has a pKa of approximately 6.0. This means that, at physiologically relevant pH values, relatively small shifts in pH will change its average charge. The unprotonated imidazole is nucleophilic and can serve as a general base, while the protonated form can serve as a general acid. In addition, it is often a ligand for transition metal ions such as iron and zinc.
- Abbreviations: His, H
- IUPAC name: 2-amino-3-(1H-imidazol-5-yl)propanoic acid
- Molecular formula: C6H9N3O2
- Molecular weight: 155.15 g/mol
lysine → lizin
Lysine is an electrically charged amino acids with basic side chains. Lysine is a base, as are arginine and histidine. The amino group is highly reactive and often participates in reactions at the active centers of enzymes. Lysine plays an important role in coordinating negatively charged ligands. It is an essential amino acid, which means that humans cannot synthesize it, so it must be ingested.
- Abbreviations: Lys, K
- IUPAC name: 2,6-diaminohexanoic acid
- Molecular formula: C6H14N2O2
- Molecular weight: 146.19 g/mol
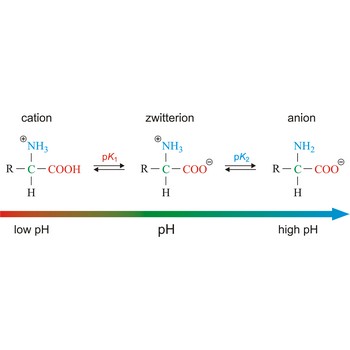
zwitterion → dipolarni ion
Zwitterion, also known as inner salt or dipolar ion, is an ion with a positive and a negative electrical charge at different locations within a molecule. As the molecule contains two opposite charges, it is electrically neutral. The term zwitterion is derived from the German word zwitter, meaning a hybrid, hermaphrodite. Zwitterions can be formed from compounds that contain both acid groups and base groups in their molecules (ampholytes).
All of the common amino acids found in proteins are ampholytes because they contain a carboxyl group (-COOH) that acts as an acid and an amino group (-NH2) that acts as a base. In the solid state, amino acids exist in the dipolar or zwitterion form. If acid is added to a solution containing the zwitterion, the carboxylate group captures a hydrogen (H+) ion, and the amino acid becomes positively charged. If base is added, ion removal of the H+ ion from the amino group of the zwitterion produces a negatively charged amino acid.
acetal → acetal
Acetals are organic compounds having the structure R2C(OR’)2 (R’ ≠ H). They are organic compounds formed by addition of alcohol molecules to aldehyde or ketone molecules. Originally, the term was confined to derivatives of aldehydes (one R = H), but it now applies equally to derivatives of ketones (neither R = H ). Mixed acetals have different R’ groups. The formation of acetals is reversible; acetals can be hydrolysed back to aldehydes (ketone) in acidic solutions.
Acetal, 1,1-diethoxyethane (CH3CH(OC2H5)2), is an organic compound, pleasant smelling, formed by addition of ethyl alcohol to ethanal (acetaldehyde). It is used as a solvent and in synthetic organic chemistry.
Citing this page:
Generalic, Eni. "Kiselo-bazna titracija." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table