structural isomer → strukturni izomer
Structural isomers are compounds that have the same formula, but different constituents. Dimethyl ether (CH3OCH3) and ethanol (CH3CH2OH), for example, are constitutional isomers with general formula C3H6O.
anomer → anomer
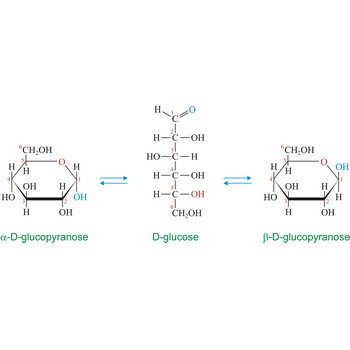
Anomers are diastereoisomers of cyclic forms of sugars or similar molecules differing in the configuration at the anomeric carbon (C-1 atom of an aldose or the C-2 atom of a 2-ketose). The cyclic forms of carbohydrates can exist in two forms, α- and β- based on the position of the substituent at the anomeric center. Anomer are designated α if the configuration at the anomeric carbon is the same as that at the reference asymmetric carbon in a Fischer projection. If the configuration differs the anomer is designated β. For example, α-D-glucopyranose and β-D-glucopyranose, the two cyclic forms of glucose, are anomers.
chiral molecule → kiralne molekule
Chiral molecule is a molecule which cannot be superimposed on its mirror image. A common example is an organic molecule containing a carbon atom to which four different atoms or groups are attached. Such molecules exhibit optical activity, i.e., they rotate the plane of a polarised light beam.
conformation → konformacija
Conformation is one of the very large numbers of possible spatial arrangements of atoms that can be interconverted by rotation about a single bond in a molecule. The conformation of a molecule is not fixed, though one or another shape may be more likely to occur. There are two extreme cases:
Staggered conformation (antiperiplanar) is a conformation about a carbon-carbon single bond in which the atoms on one carbon are as far apart as possible from the atoms on an adjacent carbon.
Eclipsed conformation (syn-periplanar) is a conformation about a carbon-carbon single bond in which the atoms on one carbon are as close as possible to the atoms on an adjacent carbon.
racemic mixture → racematna smjesa
Racemic mixture is a mixture of dextrorotatory and levorotatory optically active isomers in equal amounts. Because the two enantiomers rotate plane-polarised light in opposite directions, a racemic mixture does not rotate plane-polarised light. These mixtures are prefixed by ± or dl-.
enantiomer → enantiomer
Enantiomers are a chiral molecule and its non-superposable mirror image. The two forms rotate the plane of polarised light by equal amounts in the opposite directions. Also called optical isomers.
monosaccharide → monosaharid
Monosaccharides are carbohydrates, with the general formula Cn(H2O)n, that cannot be decomposed to a simpler carbohydrates by hydrolysis.
Depending on whether the molecule contains an aldehyde group (-CHO) or a ketone group (-CO-) monosaccharide can be a polyhydroxy aldehyde (aldose) or a polyhydroxy ketone (ketose). These aldehyde and ketone groups confer reduction properties on monosaccharides. They are also classified according to the number of carbon atoms they contain: trioses have three carbon atoms, tetroses four, pentoses five, hexoses six, heptoses seven, etc. These two systems of classification are often combined. For example, a six-carbon polyhydroxy aldehyde such as D-glucose is an aldohexose, whereas a six-carbon polyhydroxy ketone such as D-fructose is a ketohexose.
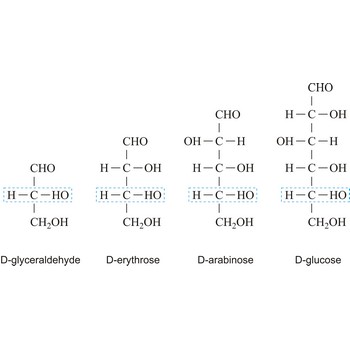
The notations D and L are used to describe the configurations of carbohydrates. In Fischer projections of monosaccharides, the carbonyl group is always placed on top (in the case of aldoses) or as close to the top as possible (in the case of ketoses). If the OH group attached to the bottom-most asymmetric carbon (the carbon that is second from the bottom) is on the right, then the compound is a D-sugar. If the OH group is on the left, then the compound is an L-sugar. Almost all sugars found in nature are D-sugars.
Monosaccharides can exist as either straight-chain or ring-shaped molecules. During the conversion from straight-chain form to cyclic form, the carbon atom containing the carbonyl oxygen, called the anomeric carbon, becomes a chiral center with two possible configurations (anomers), α and β. When the stereochemistry of the first carbon matches the stereochemistry of the last stereogenic center the sugar is the α-anomer when they are opposite the sugar is the β-anomer.
racemisation → racemizacija
Racemisation is a conversion, by heat or by chemical reaction, of an optically active compound into an optically inactive form which half of the optically active substance becomes its miror image (enantiomer).
Citing this page:
Generalic, Eni. "Izomer." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table