glycoside → glikozid
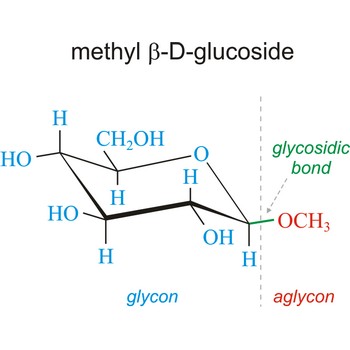
Glycoside is one of a group of organic compounds in which a sugar group is bonded through its anomeric carbon to another group via a glycosidic bond. The sugar group is known as the glycon and the non-sugar group as the aglycon. According to the IUPAC definition, all disaccharides and polysaccharides are glycosides where the aglycone is another sugar.
In the free hemiacetal form, sugars will spontaneously equilibrate between the α and β anomers. However, once the glycosidic bond is formed, the anomeric configuration of the ring is locked as either α or β. Therefore, the alpha and beta glycosides are chemically distinct. They will have different chemical, physical, and biological properties. Many glycosides occur abundantly in plants, especially as flower and fruit pigments.
The term glycoside was later extended to cover not only compounds in which the anomeric hydroxy group is replaced by a group -OR, but also those in which the replacing group is -SR (thioglycosides), -SeR (selenoglycosides), -NR1R2 (N-glycosides), or even -CR1R2R3 (C-glycosides). Thioglycoside and selenoglycoside are legitimate generic terms; however the use of N-glycoside, although widespread in biochemical literature, is improper and not recommended here (glycosylamine is a perfectly acceptable term). C-Glycoside is even less acceptable. All other glycosides are hydrolysable; the C-C bond of C-glycosides is usually not. The use and propagation of names based on C-glycoside terminology is therefore strongly discouraged.
glycosidic bond → glikozidna veza
Glycosidic bond ia a bond between the glycosyl group, the structure obtained by removing the hydroxy group from the hemiacetal function of a monosaccharide, and the -OR group (which itself may be derived from a saccharide and chalcogen replacements thereof (RS–, RSe–). The terms N-glycosides and C-glycosides are misnomers and should not be used. The glycosidic bond can be α or β in orientation, depending on whether the anomeric hydroxyl group was α or β before the glycosidic bond was formed and on the specificity of the enzymatic reaction catalyzing their formation. Once the glycosidic bond is formed, the anomeric configuration of the ring is locked as either α or β. Specific glycosidic bonds therefore may be designated α(1→4), β(1→4), α(1→6), and so on. Cellulose is formed of glucose molecules linked by β(1→4)-glycosidic bonds, whereas starch is composed of α(1→4)-glycosidic bonds.
half-life → vrijeme poluraspada
For a simple radioactive decay process, half-life, t1/2, is defined as the time required for the activity of a given radioactive isotopes to decrease to half its value by that process.
The half-life is a characteristic property of each radioactive isotope and is independent of its amount or condition.
hemiacetal → poluacetal
Hemiacetals are organic compounds having the general formula R2C(OH)OR’ (R’ ≠ H), derived from aldehydes or ketones by formal addition of an alcohol to the carbonyl group. Hemiacetals are generally unstable compounds. In some cases however, stable cyclic hemiacetals can be readily formed, especially when 5- and 6-membered rings are possible. In this case an intramolecular OH group reacts with the carbonyl group. Glucose and many other aldoses exist as cyclic hemiacetals whereas fructose and similar ketoses exist as cyclic hemiketals. Originally, the term was confined to derivatives of aldehydes (one R = H), but it now applies equally to derivatives of ketones (neither R = H).
hydration → hidratacija
Hydration is addition of water or the elements of water (i.e. H and OH) to a molecular entity. The term is also used in a more restricted sense for the process:
hydrophobic interaction → hidrofobne interakcije
Hydrophobic interaction is the tendency of hydrocarbons (or of lipophilic hydrocarbon-like groups in solutes) to form intermolecular aggregates in an aqueous medium, and analogous intramolecular interactions. The name arises from the attribution of the phenomenon to the apparent repulsion between water and hydrocarbons. Use of the misleading alternative term hydrophobic bond is discouraged.
hypsometric curve → hipsometrijska krivulja
Hypsometric curve (or hypsographic curve) shows the distribution of height of a given area (on land) and depth (at sea). The term originates from the Greek word hypsos meaning height. The part of the curve that reflects the cross section of the ocean bottom is called the bathygraphic curve.
Horizontal dashed lines indicate average height of the continents at 840 meters above sea level, and average depth of the oceans at 3 682.2 meters below sea level. If all the land above sea level (green) was moved into the sea (blue), the oceans would still be 3 km deep.
ion-product constant → ionski produkt vode
The ion-product constant. For the reaction:
the equilibrium expression would be:
Note that all pure liquid terms are omitted, hence H2O does not appear in the denominator. At 25 °C
ketal → ketal
Ketals are organic compounds formed by addition of an alcohol to a ketone. If one molecule of ketone (RR’CO) reacts with one molecule of alcohol (R"OH) then a hemiketal is formed. The rings of ketose sugars are hemiketals. Further reaction produces a full ketal (RR’C(OR")2). This term, once abandoned, has been reinstated as a subclass of acetals.
Citing this page:
Generalic, Eni. "Agreement terms and conditions analysed." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table