electrode of the third kind → elektroda trećeg reda
Electrode of the third kind is a metal electrode assembly with the equilibrium potential being a function of the concentration of a cation, other than the cation of the electrode metal, in the solution. The assembly consists of a metal in contact with two slightly soluble salts (one containing the cation of the solid metal, the other the cation to be determined, with both salts having a common anion) immersed in a solution containing a salt of the second metal (e.g., zinc metal--zinc oxalate--calcium oxalate--calcium salt solution). The potential of the metal is controlled by the concentration of its cation in the solution, but this is controlled by the anion concentration in the solution through the solubility product of the slightly soluble metal salt, which, in turn is controlled by the concentration of the cation of the second slightly soluble salt. These electrodes are very sluggish and unstable due to a series of equilibria to be established to produce a stable potential.
kelvin → kelvin
Kelvin (K) is the SI base unit of thermodynamic temperature.
The kelvin, unit of thermodynamic temperature, is the fraction 1/273.16 of the thermodynamic temperature of the triple point of water. The unit was named after the British scientist Sir. W. Thompson, Lord Kelvin (1824-1907).ketal → ketal
Ketals are organic compounds formed by addition of an alcohol to a ketone. If one molecule of ketone (RR’CO) reacts with one molecule of alcohol (R"OH) then a hemiketal is formed. The rings of ketose sugars are hemiketals. Further reaction produces a full ketal (RR’C(OR")2). This term, once abandoned, has been reinstated as a subclass of acetals.
kilogram → kilogram
Kilogram (kg) is the SI base unit of mass; it is equal to the mass of the international prototype of the kilogram.
The prototype of the standard is a cylinder of platinum-iridium alloy (90:10), 39 mm in diameter and 39 mm high. Prototype of the kilogram kept by the Bureau International des Poids et Mesures (International Bureau of Weights and Measures) at Sèevres, near Paris.
kinetic energy → kinetička energija
Kinetic energy (Ek) is associated with the state of motion of a body. It is a scalar property and defined to be
Kinetic energy is most clearly exhibited in gases, in which molecules have much greater freedom of motion than in liquids and solids.
Kirchoff, Gustav → Kirchoff, Gustav
Gustav Kirchoff (1824-1887) was a German physicist who, with the chemist Robert Bunsen (1811-1899), laid the foundations of spectral analysis. He realized that the Fraunhofer lines in the Sun's spectrum were due to light from the photosphere being absorbed at those specific wavelengths by elements in the solar atmosphere. He also found that incandescent solids, liquids, and compressed gases emit a continuous spectrum. Use of the Bunsen burner in conjunction with a glass prism led to the development of the spectroscope in collaboration with the Bunsen and to the spectroscopic discovery of the elements rubidium (1860) and cesium (1861).
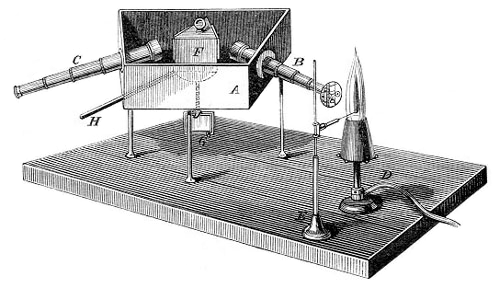
Kjeldahl flask → Kjeldahlova tikvica
Kjeldahl flask is a round bottom flask with a long wide neck that is used in the determination of nitrogen by Kjeldahl’s method. The method was developed by the Danish chemist Johan Kjeldahl (1849-1900).
Kjeldahl’s method → Kjeldahlov postupak
Kjeldahl’s method is an analytical method for determination of nitrogen in certain organic compounds. The method was developed by the Danish chemist Johan Kjeldahl (1849-1900).
It involves addition of a small amount of anhydrous potassium sulphate to the test compound, followed by heating the mixture with concentrated sulphuric acid, often with a catalyst such as copper sulphate. As a result ammonia is formed. After alkalyzing the mixture with sodium hydroxyde, the ammonia is separated by distillation, collected in standard acid, and the nitrogen determined by back-titration.
- Kjeldahl flask for decomposition (500 ml – macro or 100 ml - micro)
- funnel for alkaline solution
- Wagner tube (drop catcher)
- condenser
- absorption flask with known volume of standard acid
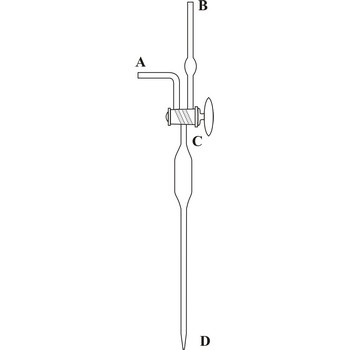
Knudsen’s pipette → Knudsenova pipeta
Kudsen's automatic pipette, developed by the Danish physicist Martin Knudsen (1871-1949), allows quick and accurate transfer of a constant volume of liquid (sea water), usually around 15 mL. On the top of pipette is a double sided C vent that can establish flow between the body of the pipette and one of the branches (A or B), or isolate the body of the pipette from both of the branches. Sucking through the B branch the pipette is filled with liquid, it is closed with a twist of the C valve and the liquid is released by rotating the valve towards the A branch (so atmospheric air can enter the pipette). Emptying the pipette takes around 30 seconds. Before it's first use, the pipette must be calibrated with distilled water.
Citing this page:
Generalic, Eni. "Alicikli�ki spojevi." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table