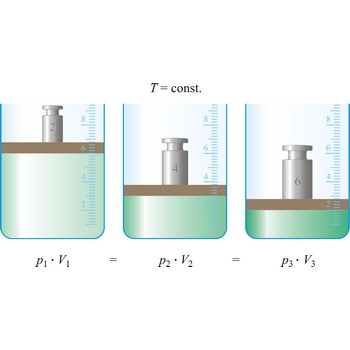
Boyle’s law → Boyleov zakon
Boyle’s law (sometimes referred to as the Boyle-Mariott’s law) is the empirical law, exact only for an ideal gas, which states that the volume of a gas is inversely proportional to its pressure at constant temperature.
Charles’ law → Charlesov zakon
The volume of a fixed mass of gas at a constant pressure expand by the constant fraction of its volume at 0 °C. For each Celsius or kelvin degree its temperature is raised. For any ideal gas fraction it is approximately 1/273. This can be expressed by the equation
were V° is the volume at 0°C and V is its volume at t°C.
This is equivalent to the statement that the volume of a fixed mass of gas at a constant pressure is proportional to its thermodynamic temperature
This law also know as Gay-Lussac’s law.
An equation similar to the one given above applies to pressures for ideal gases:
Fick’s law → Fickov zakon
Fick’s law is the statement that the flux J of a diffusing substance is proportional to the concentration gradient, i.e.,
where D is called the diffusion coefficient.
first law of thermo-dynamics → prvi zakon termodinamike
First law of thermo-dynamics is: Energy can be neither created nor destroyed, but can cross from one shape to another.
Dalton’s law → Daltonov zakon
Dalton’s law of partial pressure says that the total pressure eof gaseous mixture is equal to the sum of all gases partial pressures which make that mixture on the condition that they do not interact.
For example, if dry oxygen gas at 900 hPa is saturated with water vapor at 56 hPa, the pressure of the wet gas is 956 hPa.
law of conservation of energy → zakon o očuvanju energije
Law of conservation of energy: In an isolated system energy can be transferred from one form to another but the total energy of the system remains constant.
law of definite composition → zakon o određenom sastavu
Law of definite composition states that the elements in a given compound are always combined in the same proportion by mass. This law form the basis for the definition of a chemical compound.
Gauss’ law for electrostatics → Gaussov zakon za elektrostatiku
Gauss’ law describes the relation between charge and electric field in static situations, so it is equivalent to Coulomb’s law, which can be derived from Gauss’ law. Gauss’ law states that the net flux of electric field, Φ, through an imaginary closed surface, S, - a Gaussian surface - is equal to the net charge, q, inside that closed surface:
where electric flux Φ through Gaussian surface is given by:
ε0 is the permittivity constant and dS is a surface element.
Graham’s law → Grahamov zakon
Graham’s law is the rates at whish gases diffuse are inversely proportional to the square roots of their densities. This principle is made use of in the diffusion method of separating isotopes. The law was formulated in 1829 by British chemist Thomas Graham (1805-1869).
Citing this page:
Generalic, Eni. "Zakon o kemijskoj ravnoteži." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table