carbohydrate → ugljikohidrat
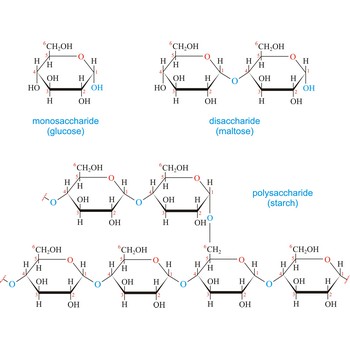
Carbohydrates (often called carbs for short) are polyhydroxy aldehydes or ketones, or substances that yield such compounds on hydrolysis. They are also known as saccharides, a term derived from the Latin word saccharum for sugar. Carbohydrates are the most abundant class of compounds in the biological world, making up more than 50 % of the dry weight of the Earth’s biomass. Every type of food we eat can have its energy traced back to a plant. Plants use carbon dioxide and water to make glucose, a simple sugar, in photosynthesis. Other carbohydrates such as cellulose and starch are made from the glucose. Light from the sun is absorbed by chlorophyll and this is converted to the energy necessary to biosynthesize carbohydrates
The term carbohydrate was applied originally to monosaccharides, in recognition of the fact that their empirical composition can be expressed as Cx(H2O)y. Later structural studies revealed that these compounds were not hydrates but the term carbohydrate persists.
Carbohydrates are generally classed as either simple or complex. Simple sugars, or monosaccharides, are carbohydrates that can’t be converted into smaller subunits by hydrolysis. Complex carbohydrates are made of two (disaccharides) or more (oligosaccharides, polysaccharides) simple sugars linked together by acetal (glycosidic) bonds and can be split into the former by hydrolysis.
combustion → gorenje
Combustion is a phenomenon when a substance is combined with oxygen in the presence of a flame accompanied by the production of heat and light. Combustion requires a supply of both fuel and oxygen (air) and can take place in the open atmosphere such as an open fire, or in a closed system, such as a car engine.
fluorescence → fluorescencija
Fluorescence is a luminescence phenomenon in which electron returns to it's ground state almost instantaneously (less than 10-8 second), and in which emission from a luminescent substance ceases when the exciting source is removed. Fluorescence is characterized by radiation emission in all directions.
geometrical optics → geometrijska optika
In most cases light can be described as an electromagnetic wave. Geometrical optics is an approximation in which the waves can be represented as straight-line rays. This approximation is valid if the light waves do not meet obstacles comparable in size to the wavelength of radiation.
chiral molecule → kiralne molekule
Chiral molecule is a molecule which cannot be superimposed on its mirror image. A common example is an organic molecule containing a carbon atom to which four different atoms or groups are attached. Such molecules exhibit optical activity, i.e., they rotate the plane of a polarised light beam.
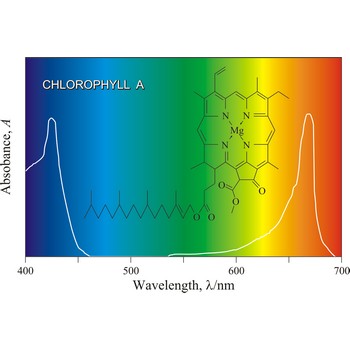
chlorophyll → klorofil
Chlorophyll is a green pigment present in green plants and cyanobacteria. Chlorophyll is essential in the transformation of light energy to chemical energy in photosynthesis. Chlorophyll absorbs light mostly in the blue and red ends of the visible spectrum, and very little in the green wavelengths. That green light is reflected, giving us the leaf colour we see.
Einstein equation → Einsteinova jednadžba
Einstein equation is the mass-energy relationship introduced by Albert Einstein in 1905 in the form E = mc2, where E is a quantity of energy, m its mass, and c is the speed of light. It presents the concept that energy possesses mass.
Einstein, Albert → Einstein, Albert
Albert Einstein (1879-1955) is a German born American physicist, who took Swiss nationality in 1901. A year later he went to work in the Bern patent office. In 1905. he published five enormously influential papers, one on Brownian movement, one on the photoelectric effect, one on the special theory of relativity, and one on energy and inertia (which included the famous expression E = mc2). In 1915 he published the general theory of relativity, concerned mainly with gravitation. In 1921 he was awarded the Nobel Prize. In 1933, as a Jew, Einstein decided to remain in the USA (where he was lecturing), as Hitler had come to power. For the remainder of his life he sought a unified field theory. In 1939 he informed president Roosevelt that an atom bomb was feasible and that. Germany might be able to make one.
Citing this page:
Generalic, Eni. "Svjetlo." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table