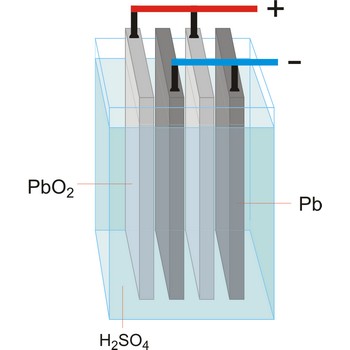
lead-acid battery → olovni akumulator
Lead-acid battery is a electrical storage device that uses a reversible chemical reaction to store energy. It was invented in 1859 by French physicist Gaston Planté. Lead-acid batteries are composed of a lead(IV) oxide cathode, a sponge metallic lead anode and a sulphuric acid solution electrolyte.
In charging, the electrical energy supplied to the battery is changed to chemical energy and stored. The chemical reaction during recharge is normally written:
In discharging, the chemical energy stored in the battery is changed to electrical energy. During discharge, lead sulfate (PbSO4) is formed on both the positive and negative plates. The chemical reaction during discharge is normally written:
Lead acid batteries are low cost, robust, tolerant to abuse, tried and tested. For higher power applications with intermittent loads however, they are generally too big and heavy and they suffer from a shorter cycle life.
octahedral molecular geometry → oktaedarska geometrija molekule
Octahedral molecular geometry (square bipyramidal shape) describes the shape of compounds where six atoms or ligands are symmetrically arranged around a central atom. The sulfur hexafluoride (SF6), with six bonding pairs, is predicted and found to be a regular octahedron. Four of the attachments are positioned in a square plane with 90° bond angles. The remaining two attachments are positioned perpendicular (90°) to the square plane at opposite ends of the central atom. Molecules with an octahedral electron pair geometries have sp3d2 (or d2sp3) hybridization at the central atom.
oil of vitriol → vitriolno ulje
Oil of vitriol is an obsolete name for sulfuric acid (H2SO4). Very old way of making sulfuric acid is by heating green vitriol (FeSO4*7H2O) to decomposition and collecting the acid vapors evolved.
organic → organski
1. Organic refers to any chemical compound based on carbon (C) with the exception of some of the simple compounds of carbon, such as carbon dioxide, which are frequently classified as inorganic compounds. Additional elements that are commonly found in organic compounds are hydrogen (H), nitrogen (N), oxygen (O), phosphorus (P) and sulfur (S).
2. Organic or organically-grown foods are grown or raised without synthetic fertilizers, pesticides, growth stimulators, or antibiotics and other drugs. Pests are controlled by cultivation techniques and the use of pesticides derived from natural sources and the use of natural fertilizers. In addition, organically grown foods must also be stored without the use of chemicals such as artificial additives and preservatives, and without food irradiation.
pickling → dekapiranje
Pickling is a process to chemically remove scale or oxide from steel to obtain a clean surface. When applied to bars or coils prior to bright drawing, the steel is immersed in a bath of dilute sulphuric acid (w(H2SO4) = 10 %) heated to a temperature of around 80 °C. An inhibitor is added to prevent attack and pitting of the cleaned metal. After pickling, a washing process takes place followed by immersion in a lime-water bath to neutralise any remaining acid.
selenium → selenij
Selenium was discovered by Jöns Jakob Berzelius (Sweden) in 1817. The origin of the name comes from the Greek word selene meaning moon. It is soft metalloid similar to sulfur. Ranges from grey metallic to red glassy appearance. Unaffected by water. Soluble in alkalis and nitric acid. Burns in air. Toxic by inhalation or ingestion. Selenium is obtained from lead, copper and nickel refining. Conducts electricity when struck by light. Light causes it to conduct electricity more easily. It is used in photoelectric cells, TV cameras, xerography machines and as a semiconductor in solar batteries and rectifiers. Also colours glass red.
silver → srebro
Silver has been known since ancient times. The origin of the name comes from the Latin word argentum meaning silver. It is silvery-ductile and malleable metal. Stable in water and oxygen. Reacts with sulfur compounds to form black sulfides. Silver is found in ores called argentite (AgS), light ruby silver (Ag3AsS3), dark ruby silver (Ag3SbS3) and brittle silver. Used in alloys for jewellery and in other compounds for photography. It is also a good conductor, but expensive.
Schrotter apparatus for determination of CO2 → Schrotterova aparatura za određivanje CO2
Schrötter decomposition apparatus (Schrötter's alkalimeter) is used to determining the carbonate content in samples of limestone, gypsum, dolomite, or baking powder by loss of weight. The apparatus is named after the Austrian chemist Anton Schrötter von Kristelli (1802-1875), who devised it in 1871. The size of the filled apparatus (apparatus is 16 cm high) is such that it weights less than 75 g, and can be placed on the pan of an analytical balance.
Procedure: Weigh about 0.5 g of the powdered carbonate sample and introduce it into the decomposition flask C. Pour into the drying tube A 2-3 mL of concentrated sulphuric acid (H2SO4), and to the dropping funnel B add about 10-15 mL of hydrochloric acid (w(HCl) = 15 %). Weigh the whole apparatus. Open the upper taps of both parts and allow the hydrochloric acid from B to run slowly down on to the powdered sample. The evolved CO2 escapes through the strong sulphuric acid and is thus thoroughly dried. When further addition of acid produces no more evolution of CO2, warm the apparatus up to 80 °C so as to expel the CO2 from the solution. Connect the upper tap of the drying tube A to a water pump and draw a slow current of air through the apparatus until completely cool. Open the upper taps for a moment to equalize the internal and external pressure and weight the apparatus again. The weight loss is equal to the weight of carbon dioxide liberated from the carbonates.
Citing this page:
Generalic, Eni. "Sumpor." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table