praseodymium → praseodimij
Praseodymium was discovered by Carl F. Auer von Welsbach (Austria) in 1885. The origin of the name comes from the Greek words prasios didymos meaning green twin. It is silvery white, moderately soft, malleable, ductile metal. Reacts slowly with oxygen. Reacts rapidly with water. Metal ignites and burns readily. Praseodymium is obtained from same salts as neodymium. Used with neodymium to make lenses for glass maker’s goggles since it filters out the yellow light present in glass blowing. Alloyed with magnesium creates a high-strength metal used in aircraft engines. Misch metal, used in the manufacture of pyrophoric alloys for cigarette lighters, contains about 5 % praseodymium metal. (Typically composition of misch metal are Ce:Nd:Pr:La:Other rare earth=50:18:6:22:4).
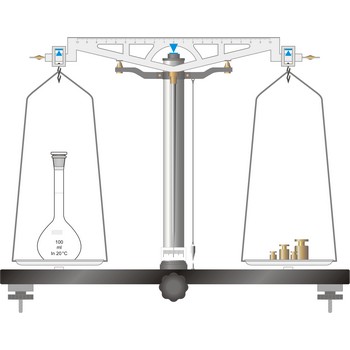
precision balance → tehnička vaga
Precision balances typically display results from three to one decimal places (0.001 g up to 0.1 g). The readability precision balances are reduced when compared to analytical balances but, precision balances accommodate higher capacities (up to several kilograms). In its traditional form, it consists of a pivoted horizontal lever of equal length arms, called the beam, with a weighing pan, also called scale, suspended from each arm.
In electronic top pan, or toploader balances, mass is determined not by mechanical deflection but by electronically controlled compensation of an electric force. The signal generated enables the mass to be read from a digital display. The mass of the empty container can be stored in the balance’s computer memory and automatically deducted from the mass of the container plus its contents.
promethium → prometij
Promethium was discovered by J. A. Marinsky, Lawrence Glendenin and Charles D. Coryell (USA) in 1945. Named after Prometheus in Greek mythology, who stole fire from the gods. It is rare earth metal of synthetic origin on the earth, naturally made in stars. Poison. Radiotoxic. Radioactive. Promethium does not occur naturally. Found among fission products of uranium, thorium and plutonium. It has been used as a source of radioactivity for thickness-measuring gages.
protactinium → protaktinij
Protactinium was discovered by Otto Hahn (Germany) and Lise Meitner (Austria) in 1917. The origin of the name comes from the Greek word protos meaning first. It is very rare, silvery-white, extremely radioactive metal. Resists alkalis; reacts with oxygen and acids. Attacked by steam. Highly radiotoxic. Protactinium is extremely toxic and must be handled with great care. Protactinium does not occur in nature. Found among fission products of uranium, thorium and plutonium.
proton → proton
Proton is a stable elementary particle of unit positive charge and spin 1/2. Protons and neutrons, which are collectively called nucleons, are the constituents of the nucleus.
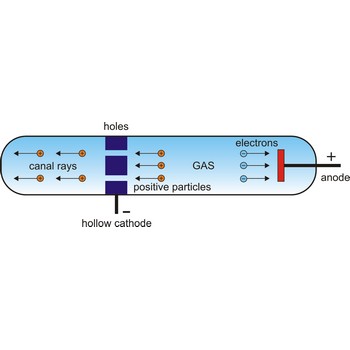
In 1886, German physicist Eugene Goldstein (1850-1930) discovered positive particles by using a modified Crookes tube with holes in the cathode in an evacuated tube. When cathode rays were given off in one direction toward the anode, other rays found their way through the holes in the cathode and sped off in the opposite direction. Since these other rays traveled in the direction opposite to the negatively charged cathode rays, it seemed that they must be composed of positively charged particles. Rutherford suggested that this fundamental positive particle be called the proton.
radium → radij
Radium was discovered by Marie and Pierre Curie (France) in 1898. The origin of the name comes from the Latin word radius meaning ray. It is silvery-white radioactive metal. Reacts with oxygen and water. Highly radiotoxic. Carcinogen by inhalation, ingestion, or exposure. Radium is found in uranium ores at 1 part per 3 million parts uranium. Used in treating cancer because of the gamma rays it gives off.
radon → radon
Radon was discovered by Friedrich Ernst Dorn (Germany) in 1900. The origin of the name is variation of the name of element radium; radon was called niton at first, from the Latin word nitens meaning shining. It is colourless, odourless radioactive, heavy, noble gas. Chemically inert and non-flammable. Highly radiotoxic. Carcinogen by inhalation. Radon is formed from the decay of radium in the earths crust. Used to treat some forms of cancer.
Rayleigh number → Rayleighova značajka
Rayleigh number (Ra) is a dimensionless quantity used in fluid mechanics, defined by
where l is length, g is acceleration of gravity, α is cubic expansion coefficient, T is temperature, ρ is density, η is viscosity, and a is thermal diffusivity.
redox reaction → redoks-reakcija
Redox reaction is an oxidation-reduction reaction is a reaction in which one or more electrons are transferred. When an atom, ion, or molecule loses one or more electrons, it is oxidised. When an atom, ion, or molecule gains one or more electrons, it is reduced.
reducing agent → redukcijsko sredstvo
Reducing agent may be defined in various ways, depending upon the context in which the phrase is used. In broad terms it is often taken to mean a chemical which can act as an electron donor. Thus, in the reaction:
the zinc is being reduced (gaining electrons) by reaction with the iron cations; the Fe2+ in this instance is acting as a reducing agent.
Citing this page:
Generalic, Eni. "Ledište." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table