supersaturated solution → prezasićena otopina
Solution is supersaturated when it contains greater quantity of dissolved substance in itself than it corresponds to solubility of that substance at that temperature. It is said to be in an unstable state, and by shaking the vessel containing that such a solution separation of salt surplus can occur.
Hesse’s law → Hessov zakon
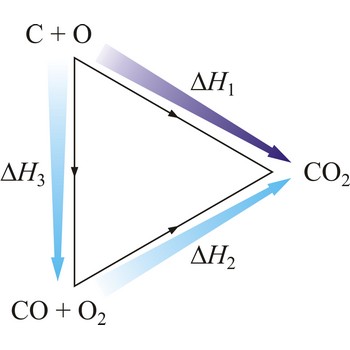
Hesse’s law says that reaction heat of some chemical change does not depend on the way in which the reaction is conducted, but only on starting and ending system state. Hesse’s law is also known as the law of constant heat summation. Hesse’s law is also known as the law of constant heat summation. The law was first put forward in 1840 by the Swiss-born Russian chemist Germain Henri Hess (1802-1850).
Hesse’s law can be used to obtain thermodynamic data that cannot be measured directly. For example, it is very difficult to control the oxidation of graphite to give pure CO. However, enthalpy for the oxidation of graphite to CO2 can easily be measured. So can the enthalpy of oxidation of CO to CO2. The application of Hess’s law enables us to estimate the enthalpy of formation of CO.
| C(s) + O2(g) →← CO2(g) | ΔrH1 = -393 kJ mol-1 |
| CO(g) + 1/2O2(g) →← CO2(g) | ΔrH2 = -283 kJ mol-1 |
| C(s) + 1/2O2(g) →← CO(g) | ΔrH3 = -110 kJ mol-1 |
The equation shows the standard enthalpy of formation of CO to be -110 kJ/mol.
high fructose corn syrup → visoko fruktozni kukuruzni sirup
High fructose corn syrup (HFCS) is commonly used in place of sugar in foods and drinks. Corn starch is hydrolyzed to glucose, which is then treated with glucose isomerase to produce a fructose-rich mixture. HFCS is available in a number of forms, named according to the percentage of fructose they contain, HFCS-55 for instance contains 55 % fructose and 45 % glucose. The enhanced sweetness, low cost and ease of use are the main reasons why manufacturers now prefer to use high fructose corn syrup instead of sugar.
surfactant → površinski aktivna tvar
Surfactants are substances which diminish the surface tension of liquids.
system → sustav
System is the region under consideration, as distinguished from the rest of the universe (the environment). Systems may be separated from environments by boundaries that prevent the transfer of mass (a closed system), of heat (an adiabatic system), or of any energy (an isolated system). Systems that exchange mass with the environment are open systems.
histidine → histidin
Histidine is an electrically charged amino acids with basic side chains. It is an essential amino acid, which means that humans cannot synthesize it, so it must be ingested. Histidine is perhaps the most common and versatile catalytic residue in proteins. The imidazole sidechain of histidine has a pKa of approximately 6.0. This means that, at physiologically relevant pH values, relatively small shifts in pH will change its average charge. The unprotonated imidazole is nucleophilic and can serve as a general base, while the protonated form can serve as a general acid. In addition, it is often a ligand for transition metal ions such as iron and zinc.
- Abbreviations: His, H
- IUPAC name: 2-amino-3-(1H-imidazol-5-yl)propanoic acid
- Molecular formula: C6H9N3O2
- Molecular weight: 155.15 g/mol
holmium → holmij
Holmium was discovered by Per Theodore Cleve (Sweden) in 1879. The origin of the name comes from the Greek word Holmia meaning Stockholm. It is fairly soft, malleable, lustrous, silvery metal. Reacts slowly with oxygen and water. Dissolves in acids. Can react violently with air or halogens. Holmium occurs in gadolinite. Most often from monazite sand. It has very few practical applications; however, it has some unusual magnetic properties that offer some hope for future applications.
homologous series → homologni niz
Series of compounds which have a common general formula and in which each member differs from the next member by a constant unit, which is the methylene group (-CH2-) is called the homologous series. Members of a homologous series are called homolog.
An example of the homologous series with some of their homologs are given below. Straight chain alkanes having general formula CnH2n+2
| Structure | Name |
|---|---|
| CH4 | methane |
| CH3-CH3 | ethane |
| CH3-CH2-CH3 | propane |
| CH3-CH2CH2CH3 | butane |
| CH3-(CH2)3-CH3 | pentane |
| CH3-(CH2)4-CH3 | hexane |
| CH3-(CH2)5-CH3 | heptane |
| CH3-(CH2)6-CH3 | octane |
| CH3-(CH2)7-CH3 | nonane |
| CH3-(CH2)8-CH3 | decane |
hydration → hidratacija
Hydration is addition of water or the elements of water (i.e. H and OH) to a molecular entity. The term is also used in a more restricted sense for the process:
hydrogen → vodik
Hydrogen was discovered by Sir Henry Cavendish (England) in 1766. The origin of the name comes from the Greek words hydro and genes meaning water and generate. It is colourless, odourless gas, burns and forms explosive mixtures in air. Reacts violently with oxidants. Hydrogen is the most abundant element in the universe. Commercial quantities of hydrogen are produced by reacting superheated steam with methane or carbon. In lab work from reaction of metals with acid solutions or electrolysis. Most hydrogen is used in the production of ammonia and in metal refining. Also used as fuel in rockets. Its two heavier isotopes (deuterium and tritium) used respectively for nuclear fusion.
Citing this page:
Generalic, Eni. "Ledište." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table