quasicrystal → kvazikristal
Quasicrystal is a solid having conventional crystalline properties but whose lattice does not display translational periodicity.
Daniell cell → Daniellov članak
In 1836 the British chemist John Frederic Daniell (1790-1845) proposed an improved electric cell that supplied an even current during continuous operation. Daniell cell consisted of a glass jar containing copper and zinc electrodes, each immersed in their respective acidic sulphate solutions. The two solutions were separated by a porous clay cylinder separator. It was a galvanic cell in which the spontaneous electrodissolution of zinc and electroplating of copper provided the electrical current.
Zn(s) |
→ | Zn2+ + 2e- |
+0.763 V |
Cu2+ + 2e- |
→ | Cu(s) |
+0.337 V |
Zn(s) + Cu2+ |
→← | Zn2+ + Cu(s) |
+1.100 V |
deionised water → deionizirana voda
Deionised water is water from which ionic salts have been removed by ion-exchange. It is used for many purposes as an alternative to distilled water.
| Type of water | Conductivity / µScm-1 |
|---|---|
| Ultrapure water | 0.05 |
| Distilled water | 0.5 |
| Tap water | 50 |
| Ocean water | 50 000 |
dielectric constant → dielektrična konstanta
Dielectric constant or permittivity (ε) is an index of the ability of a substance to attenuate the transmission of an electrostatic force from one charged body to another. The lower the value, the greater the attenuation. The standard measurement apparatus utilises a vacuum whose dielectric constant is 1. In reference to this, various materials interposed between the charged terminal have the following value at 20 °C:
| vacuum | 1 |
| air | 1.00058 |
| glass | 3 |
| benzene | 2.3 |
| acetic acid | 6.2 |
| ammonia | 15.5 |
| ethanol | 25 |
| glycerol | 56 |
| water | 81 |
The exceptionally high value for water accounts for its unique behaviour as a solvent and in electrolytic solutions. Dielectric constant values decrease as the temperature rises.
disaccharide → disaharid
Disaccharides are compounds in which two monosaccharides are joined by a glycosidic bond. A glycosidic bond to the anomeric carbon can be either α or β. For example, maltose, the disaccharide obtained by enzyme-catalyzed hydrolysis of starch, consists of two D-glucopyranose units joined by a 1,4’-α-glycoside bond. The "prime" superscript indicates that C-4 is not in the same ring as C-1. Unlike the other disaccharides, sucrose is not a reducing sugar and does not exhibit mutarotation because the glycosidic bond is between the anomeric carbon of glucose and the anomeric carbon of fructose.
Earth’s crust → Zemljina kora
Crust is outer layer of the solid earth, above the Mohorovicic discontinuity. Its thickness averages about 35 km on the continents and about 7 km below the ocean floor, and has the approximate chemical composition:
| Element | Percentage (%) |
|---|---|
| oxygen | 47 |
| silicon | 28 |
| aluminium | 8 |
| iron | 4.5 |
| calcium | 3.5 |
| sodium | 2.5 |
| potassium | 2.5 |
| magnesium | 2.2 |
fructose → fruktoza
Fructose (fruit sugar) is a ketohexose (a six-carbon ketonic sugar), which occurs in sweet fruits and honey. Glucose and fructose have the same molecular formula, C6H12O6, but have different structures. Pure, dry fructose is a very sweet, white, odorless, crystalline solid. Fructose is one of the sweetest of all sugars and is combined with glucose to make sucrose, or common table sugar. An older common name for fructose is levulose, after its levorotatory property of rotating plane polarized light to the left (in contrast to glucose which is dextrorotatory). The polysaccharide inulin is a polymer of fructose.
homologous series → homologni niz
Series of compounds which have a common general formula and in which each member differs from the next member by a constant unit, which is the methylene group (-CH2-) is called the homologous series. Members of a homologous series are called homolog.
An example of the homologous series with some of their homologs are given below. Straight chain alkanes having general formula CnH2n+2
| Structure | Name |
|---|---|
| CH4 | methane |
| CH3-CH3 | ethane |
| CH3-CH2-CH3 | propane |
| CH3-CH2CH2CH3 | butane |
| CH3-(CH2)3-CH3 | pentane |
| CH3-(CH2)4-CH3 | hexane |
| CH3-(CH2)5-CH3 | heptane |
| CH3-(CH2)6-CH3 | octane |
| CH3-(CH2)7-CH3 | nonane |
| CH3-(CH2)8-CH3 | decane |
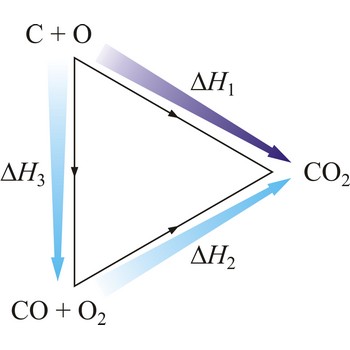
Hesse’s law → Hessov zakon
Hesse’s law says that reaction heat of some chemical change does not depend on the way in which the reaction is conducted, but only on starting and ending system state. Hesse’s law is also known as the law of constant heat summation. Hesse’s law is also known as the law of constant heat summation. The law was first put forward in 1840 by the Swiss-born Russian chemist Germain Henri Hess (1802-1850).
Hesse’s law can be used to obtain thermodynamic data that cannot be measured directly. For example, it is very difficult to control the oxidation of graphite to give pure CO. However, enthalpy for the oxidation of graphite to CO2 can easily be measured. So can the enthalpy of oxidation of CO to CO2. The application of Hess’s law enables us to estimate the enthalpy of formation of CO.
| C(s) + O2(g) →← CO2(g) | ΔrH1 = -393 kJ mol-1 |
| CO(g) + 1/2O2(g) →← CO2(g) | ΔrH2 = -283 kJ mol-1 |
| C(s) + 1/2O2(g) →← CO(g) | ΔrH3 = -110 kJ mol-1 |
The equation shows the standard enthalpy of formation of CO to be -110 kJ/mol.
hydrosphere → hidrosfera
Hydrosphere (from the Greek for water sphere) is a discontinuous layer of water on, under, and over the Earth's surface. It includes all liquid and frozen surface waters, groundwater held in soil and rock, and atmospheric water vapour. Water continuously circulates between these reservoirs in what is called the hydrologic cycle, which is driven by energy from the Sun.
| Reservoir | V / 106 km3 | w / % |
|---|---|---|
| oceans | 1 370.0 | 97.25 |
| ice caps and glaciers | 29.0 | 2.05 |
| groundwater | 9.5 | 0.68 |
| lakes, rivers | 0.127 | 0.01 |
| soil moisture | 0.065 | 0.005 |
| atmosphere (as liquid equivalent of water vapour) | 0.013 | 0.001 |
| biosphere | 0.0006 | 0.00004 |
| TOTAL | 1 408.7 | 100 |
Citing this page:
Generalic, Eni. "La primer tabla periodica." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table