fuel cell → gorivi članak
Fuel cell is a device that converts chemical energy into electrical energy. It is different from a battery in that the energy conversion continues as long as fuel and oxidising agent are fed to the fuel cell; that is, in principle indefinitely. (A battery is manufactured with a limited amount of chemicals, and it is exhausted when all the chemicals have reacted.) It is a galvanic cell where spontaneous chemical reactions occur at the electrodes. The fuel is oxidised at the anode, and the oxidising agent (almost always oxygen or air) is reduced at the cathode. Presently, the most commonly used fuel is hydrogen. More conventional fuels (e.g., petrol or natural gas) must be converted (reformed) into hydrogen before they can be utilised in a fuel cell.
Some fuel cells employ an aqueous solution as electrolyte, that can be either acidic or basic (alkaline), or an ion-exchange membrane soaked in aqueous solution can act as the electrolyte. These fuel cells operate at relatively low temperatures (from room temperature to not much above the boiling point of water). Some fuel cells employ molten salts (especially carbonates) as electrolytes and have to operate at temperatures of several hundred degrees centigrade (Celsius). Others employ ionically conductive solids as electrolyte and must operate close to 1 000 °C.
lead → olovo
Lead has been known since ancient times. The origin of the name comes from the Latin word plumbum meaning liquid silver. It is very soft, highly malleable and ductile, blue-white shiny metal. Tarnishes in moist air; stable in oxygen and water. Dissolves in nitric acid. Compounds toxic by inhalation or ingestion. Danger of cumulative effects. Lead is found most often in ores called galena or lead sulfide (PbS). Used in solder, shielding against radiation and in batteries.
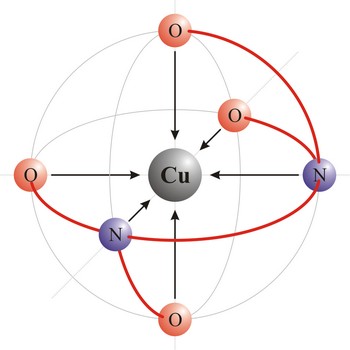
ligand → ligand
Ligand is an ion (F-, Cl-, Br-, I-, S2-, CN-, NCS-, OH-, NH2-) or molecule (NH3, H2O, NO, CO) that donates a pair of electrons to a metal atom or ion in forming a coordination complex. The main way of classifying ligands is by the number of points at which they are attached to, or bound to, the metal center. This is the denticity. Ligands with one potential donor atom are monodentate. Polydentate ligand is a ligand that is attached to a central metal ion by bonds from two or more donor atoms. Ligands with more than one potential donor atom are known as ambidentate, such as the thiocyanate ion, NCS-, which can bind to the metal center with either the nitrogen or sulphur atoms. Chelating ligands are those polydentate ligands which can form a ring including the metal atom.
limestone → vapnenac
Limestone is a sedimentary rock composed primarily of calcium carbonate in the form of the mineral calcite.. Some 10 % to 15 % of all sedimentary rocks are limestones. Limestone is usually organic, but it may also be inorganic. Calcium carbonate may have been directly precipitated from the sea-water or by the lithification of coral reefs, marine organism shells, or marine organism skeletons.
mineral → mineral
Minerals are compounds in which metals can be found in nature. Metals in nature can appear as:
| autochthonous | Au, Cu, Pt, Ag, Pd, Hg, Ir |
| oxides | Fe, Al, Sn, Cr, Mn, W, Cu |
| sulphides | Cu, Pb, Zn, Ni, Ag, Co, Sb, Hg, Mo, Cd, Bi |
| carbonates | Fe, Zn, Cu, Mg, Mn, Pb |
| silicates | Ni, Cu, Zn, Mn |
| chlorides | Ag, Cu, Mg, Na, K |
| sulphates | Ca, Ba, Sr, Cu |
organic chemistry → organska kemija
Organic chemistry is a science about all compounds which consist of carbon atoms, except carbonates and oxides of carbon.
polariser → polarizator
Polariser is frequently a specially made prism from crystal of Iceland Spar (form of calcium carbonate), also known as Nicol’s prism. It can also be a small plate of tourmaline.
Citing this page:
Generalic, Eni. "Karbonati." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table