ion-product constant → ionski produkt vode
The ion-product constant. For the reaction:
the equilibrium expression would be:
Note that all pure liquid terms are omitted, hence H2O does not appear in the denominator. At 25 °C
iron → željezo
Iron has been known since ancient times. The origin of the name comes from the Latin word ferrum meaning iron. It is malleable, ductile, silvery-white metal. Exposed surfaces form red-brown oxides. Forms very strong alloys (steel). Ferromagnetic. Metal dust flammable. Fourth most abundant element in the earth’s crust. Iron is obtained from iron ores. Pure metal produced in blast furnaces by layering limestone, coke and iron ore and forcing hot gasses into the bottom. This heats the coke red hot and the iron is reduced from its oxides and liquefied where it flows to the bottom. Iron is the most common metal in human society. More than 90 % of all metal refined in the world is iron. Used in steel and other alloys. It is the chief constituent of hemoglobin which carries oxygen in blood vessels. Its oxides are used in magnetic tapes and disks.
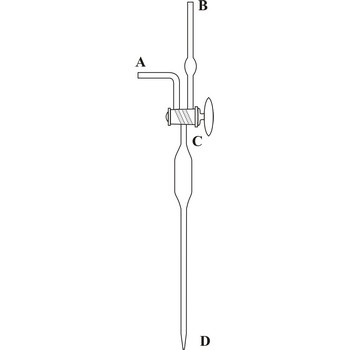
Knudsen’s pipette → Knudsenova pipeta
Kudsen's automatic pipette, developed by the Danish physicist Martin Knudsen (1871-1949), allows quick and accurate transfer of a constant volume of liquid (sea water), usually around 15 mL. On the top of pipette is a double sided C vent that can establish flow between the body of the pipette and one of the branches (A or B), or isolate the body of the pipette from both of the branches. Sucking through the B branch the pipette is filled with liquid, it is closed with a twist of the C valve and the liquid is released by rotating the valve towards the A branch (so atmospheric air can enter the pipette). Emptying the pipette takes around 30 seconds. Before it's first use, the pipette must be calibrated with distilled water.
lanthanides → lantanoidi
Lanthanides (lanthanons, lanthanoids or rare-earth elements) are a series of fourteen elements in the periodic table, generally considered to range in proton number from cerium to lutetium inclusive. It was convenient to divide these elements into the cerium group or light earth: cerium (Ce), praseodymium (Pr), neodymium (Nd), promethium (Pm), samarium (Sm), europium (Eu); and the yttrium group or heavy earths: gadolinium (Gd), terbium (Tb), dysprosium (Dy), holmium (Ho), erbium (Er), thulium (Tm), ytterbium (Yb) i lutetium (Lu). The position of lanthanum is somewhat equivocal and, although not itself a lanthanide, it is often included with them for comparative purpose. The lanthanides are sometimes simply called the rare earths. Apart from unstable Pm, the lanthanides are actually not rare. Cerium is the 26. most abundant of all elements, 5 times as abundant as Pb. All are silvery very reactive metals.
lead → olovo
Lead has been known since ancient times. The origin of the name comes from the Latin word plumbum meaning liquid silver. It is very soft, highly malleable and ductile, blue-white shiny metal. Tarnishes in moist air; stable in oxygen and water. Dissolves in nitric acid. Compounds toxic by inhalation or ingestion. Danger of cumulative effects. Lead is found most often in ores called galena or lead sulfide (PbS). Used in solder, shielding against radiation and in batteries.
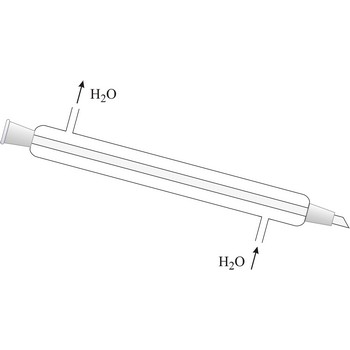
Liebig condenser → Liebigovo hladilo
Liebig condenser is used for condensing of vapours that pass trough the centre tube. It is cooled with water that passes in the outer tube (shell around the centre tube) in the opposite direction than the one of hot vapour. Though named after the German chemist Justus von Liebig (1803-1873), he cannot be given credit for having invented it because it had already been in use for some time before him.
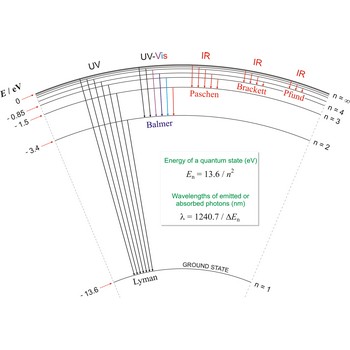
Lyman series → Lymanova serija
Lyman series is the series of lines in the spectrum of the hydrogen atom which corresponds to transitions between the ground state (principal quantum number n = 1) and successive excited states.
magnetic permeability → magnetska permeabilnost
Magnetic permeability (μ), also called permeability, is a constant of proportionality that exists between magnetic induction and magnetic field intensity. This constant is equal to approximately μo = 1.257×10-6 H/m in a vacuum.
Magnetic permeability is often expressed in relative, rather than in absolute, terms. If μ represents the permeability of the substance in question, then the relative permeability, μr, is given by:
mercury → živa
Mercury has been known since ancient times. The origin of the name comes from the Latin word hydrargyrum meaning liquid silver. It is heavy, silver-white metal, liquid at ordinary temperatures. Stable in air and water. Unreactive with alkalis and most acids. Gives off poisonous vapour. Chronic cumulative effects. Mercury only rarely occurs free in nature. The chief ore is cinnabar or mercury sulfide (HgS). Used in thermometers, barometers and batteries. Also used in electrical switches and mercury-vapour lighting products.
metal → metal
Metals are materials in which the highest occupied energy band (conduction band) is only partially filled with electrons.
Their physical properties generally include:
- They are good conductors of heat and electricity. The electrical conductivity of metals generally decreases with temperature.
- They are malleable and ductile in their solid state.
- They show metallic lustre.
- They are opaque.
- They have high density.
- They are solids (except mercury)
- They have a crystal structure in which each atom is surrounded by eight to twelve near neighbours
Their chemical properties generally are:
- They have one to four valence electrons.
- They have low ionisation potentials; they readily lose electrons.
- They are good reducing agents.
- They have hydroxides which are bases or amphoteric.
- They are electropositive.
Metallic characteristics of the elements decrease and non-metallic characteristics increase with the increase of valence electrons. Also metallic characteristics increase with the number of electron shells. Therefore, there is no sharp dividing line between the metals and non-metals.
Of the 114 elements now known, only 17 show primarily non-metallic characteristics, 7 others are metalloids, and 89 may be classed as metals.
Citing this page:
Generalic, Eni. "Having such a good time with me." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table