alkaline earth metal → zemnoalkalijski metal
Alkali earth metal is a term that refers to six elements: beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra). These elements make up group 2 of the periodic table of elements. They all exhibit a single oxidation state, +2. They are all light and very reactive. Barium and radium are the most reactive and beryllium is the least.
To denote slightly soluble metal oxides chemists formerly used the term "earth". The oxides of barium, strontium, and calcium resemble alumina (Al2O3), a typical "earth", but form alkaline mixtures with water. For this reason barium, strontium, and calcium were called alkaline earth metals. This name has now been extended to include all of the elements of group 2.
diatomaceous earth → dijatomejska zemlja
Diatomaceous earth is a naturally occurring siliceous sedimentary mineral compound from microscopic skeletal remains (frustules) of diatoms, unicellular aquatic plants of microscopic size. Their fossilized remains are called diatomite and contains approximately 3000 diatom frustules per cubic millimetre.
Diatomite is relatively inert and has a high absorptive capacity, large surface area, and low bulk density. It consists of approximately 90 % silica, and the remainder consists of compounds such as aluminum and iron oxides. The fine pores in the diatom frustules make diatomite an excellent filtering material for waters, beverages, oils, chemicals, as well as many other products.
Earth’s crust → Zemljina kora
Crust is outer layer of the solid earth, above the Mohorovicic discontinuity. Its thickness averages about 35 km on the continents and about 7 km below the ocean floor, and has the approximate chemical composition:
| Element | Percentage (%) |
|---|---|
| oxygen | 47 |
| silicon | 28 |
| aluminium | 8 |
| iron | 4.5 |
| calcium | 3.5 |
| sodium | 2.5 |
| potassium | 2.5 |
| magnesium | 2.2 |
rare earth elements → elementi rijetkih zemalja
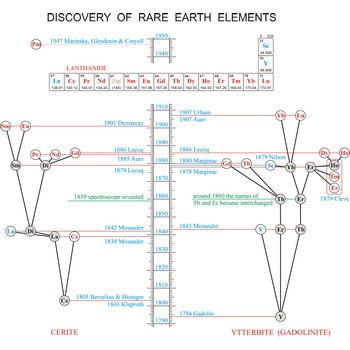
Rare earth elements (metals) are the elements scandium (Sc), yttrium (Y), and the lanthanides (La, Ce, Pr, Nd, Pm, Sm, Eu, Gd, Tb, Dy, Ho, Er, Tm, Yb, Lu). These elements got their name from the fact that chemists first isolated them in their oxide forms. These oxides somewhat resemble calcium, magnesium and aluminium oxides, sometimes called common earths. Do you want to know more?
ecological footprint → ekološki otisak
The Ecological Footprint is defined as the area of productive land and water ecosystems required to produce the resources that the population consumes (food, fiber, timber, energy, and space for infrastructure) and assimilate the wastes that the population produces (CO2 is the only waste product currently included), wherever on Earth the land and water is located. It compares actual throughput of renewable resources relative to what is annually renewed. Non-renewable resources are not assessed, as by definition their use is not sustainable.
Ecological footprints and biocapacity are expressed in global hectares (gha). Each unit corresponds to one hectare of biologically productive space with world average productivity. In U.S. Footprint results are often presented in global acres (ga). One U.S. acre is equal to 0.405 hectares.
Humanity is currently consuming renewable resources at a faster rate than ecosystems can regenerate them and continuing to release more CO2 than ecosystems can absorb. In 2007, humanity's Footprint was 18 billion gha, or 2.7 gha per person. However, the Earth's biocapacity was only 11.9 billion gha, or 1.8 gha per person. This represents an ecological overshoot of 50 per cent. Put another way, people used the equivalent of 1.5 planets to support their activities (more developed countries generally make higher demands on the Earth's ecosystems than poorer, less developed countries).
abundance of elements → rasprostranjenost elemenata
Elements in nature are mostly found in different compounds and, rarely, in the free (elementary) state. In Earth’s crust the most abundant of all elements is oxygen (with 49.5 %), then silicon (25 %), aluminium (7.5 %), iron (4.7 %), calcium (3.4 %), sodium (2.6 %), potassium (2.4 %), magnesium (1.9 %) and hydrogen (1.9 %). These nine elements make up almost 99 % of the Earth’s composition.
abundance of substances → rasprostranjenost tvari
Abundance of substances is the ratio of the total mass of a specified element in the Earth’s crust to the total mass of the Earth’s crust. It is often expressed as a percentage.
aluminium → aluminij
Aluminium was discovered by Friedrich Wöhler (Germany) in 1827. The origin of the name comes from the Latin word alumen meaning alum. It is soft, lightweight, silvery-white metal. Exposed surfaces quickly form protective oxide coating. Metal reacts violently with oxidants. Third most abundant element in the earth’s crust. Aluminium is the most abundant metal to be found in the earth’s crust, but is never found free in nature. Aluminium is obtained by electrolysis from bauxite. Used for many purposes from airplanes to beverage cans. Too soft in its pure form so less than 1 % of silicon or iron is added, which hardens and strengthens it.
astronomical unit → astronomska jedinica
Astronomical unit (AU) is a unit of length employed in astronomy for describing planetary distance. It is the mean distance of the earth from the sun, equal to 1.49597870×1011 m.
atmospheric pressure → atmosferski tlak
Atmospheric pressure is the pressure exerted by weight of the air above it at any point on the earth’s surface. At sea level the atmosphere will support a column of mercury about 760 mm high. This decreases with increasing altitude. The standard value for the atmospheric pressure at sea level in SI units is 101 325 Pa.
Citing this page:
Generalic, Eni. "Earth overshoot day." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table