gravimetry → gravimetrija
Gravimetry is the quantitative measurement of an analyte by weighing a pure, solid form of the analyte. Since gravimetric analysis is an absolute measurement, it is the principal method for analysing and preparing primary standards.
A typical experimental procedure to determine an unknown concentration of an analyte in a solution is as follows:
- quantitatively precipitate the analyte from the solution
- collect the precipitate by filtering and wash it to remove impurities
- dry the solid in an oven to remove the solvent
- weigh the solid on an analytical balance
- calculate the analyte concentration in the original solution based on the weight of the precipitate.
hafnium → hafnij
Hafnium was discovered by Dirk Coster (Denmark) and Georg Karl von Hevesy (Hungary) in 1923. The origin of the name comes from the Latin name Hafnia meaning Copenhagen. It is silvery, ductile metal. Exposed surfaces form oxide film. Resists alkalis and acids (except HF). Toxic. Metal ignites and burns readily. Hafnium is obtained from mineral zircon or baddeleyite. Used in reactor control rods because of its ability to absorb neutrons.
hassium → hassij
Hassium was discovered by Peter Armbruster, Gottfried Münzenber and their co-workers at the Heavy Ion Research Laboratory (Gesellschaft für Schwerionenforschung, GSI) in Darmstadt, Germany in 1984. The origin of the name is the Latin word Hassias meaning Hess, the German state. It is synthetic radioactive metal. Hassium was produced bythe bombardment of lead-208 with iron-58.
helium → helij
Helium was discovered by Pierre Jules César Janssen (France) and Sir William Ramsay (Scotland) in 1868. The origin of the name comes from the Greek word helios meaning sun. It is light, odourless, colourless inert gas. Second most abundant element in the universe. Helium is found in natural gas deposits from wells in Texas, Oklahoma and Kansas. Used in balloons, deep sea diving and welding. Also used in very low temperature research.
holmium → holmij
Holmium was discovered by Per Theodore Cleve (Sweden) in 1879. The origin of the name comes from the Greek word Holmia meaning Stockholm. It is fairly soft, malleable, lustrous, silvery metal. Reacts slowly with oxygen and water. Dissolves in acids. Can react violently with air or halogens. Holmium occurs in gadolinite. Most often from monazite sand. It has very few practical applications; however, it has some unusual magnetic properties that offer some hope for future applications.
Hesse’s law → Hessov zakon
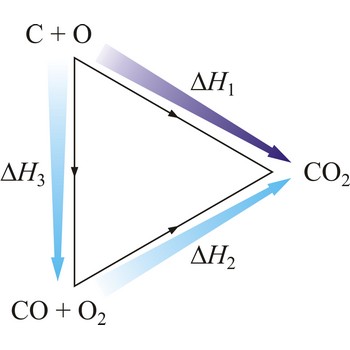
Hesse’s law says that reaction heat of some chemical change does not depend on the way in which the reaction is conducted, but only on starting and ending system state. Hesse’s law is also known as the law of constant heat summation. Hesse’s law is also known as the law of constant heat summation. The law was first put forward in 1840 by the Swiss-born Russian chemist Germain Henri Hess (1802-1850).
Hesse’s law can be used to obtain thermodynamic data that cannot be measured directly. For example, it is very difficult to control the oxidation of graphite to give pure CO. However, enthalpy for the oxidation of graphite to CO2 can easily be measured. So can the enthalpy of oxidation of CO to CO2. The application of Hess’s law enables us to estimate the enthalpy of formation of CO.
| C(s) + O2(g) →← CO2(g) | ΔrH1 = -393 kJ mol-1 |
| CO(g) + 1/2O2(g) →← CO2(g) | ΔrH2 = -283 kJ mol-1 |
| C(s) + 1/2O2(g) →← CO(g) | ΔrH3 = -110 kJ mol-1 |
The equation shows the standard enthalpy of formation of CO to be -110 kJ/mol.
hydrogen → vodik
Hydrogen was discovered by Sir Henry Cavendish (England) in 1766. The origin of the name comes from the Greek words hydro and genes meaning water and generate. It is colourless, odourless gas, burns and forms explosive mixtures in air. Reacts violently with oxidants. Hydrogen is the most abundant element in the universe. Commercial quantities of hydrogen are produced by reacting superheated steam with methane or carbon. In lab work from reaction of metals with acid solutions or electrolysis. Most hydrogen is used in the production of ammonia and in metal refining. Also used as fuel in rockets. Its two heavier isotopes (deuterium and tritium) used respectively for nuclear fusion.
hypsometric curve → hipsometrijska krivulja
Hypsometric curve (or hypsographic curve) shows the distribution of height of a given area (on land) and depth (at sea). The term originates from the Greek word hypsos meaning height. The part of the curve that reflects the cross section of the ocean bottom is called the bathygraphic curve.
Horizontal dashed lines indicate average height of the continents at 840 meters above sea level, and average depth of the oceans at 3 682.2 meters below sea level. If all the land above sea level (green) was moved into the sea (blue), the oceans would still be 3 km deep.
iodine → jod
Iodine was discovered by Bernard Courtois (France) in 1811. The origin of the name comes from the Greek word iodes meaning violet. It is shiny, black, non-metallic solid with characteristic odour. Sublimes easily and as a gas it is violet and intensely irritating to the eyes, nose and throat. Iodine occurs on land and in the sea in sodium and potassium compounds. Required in small amounts by humans. Once used as an antiseptic, but no longer due to its poisonous nature.
iridium → iridij
Iridium was discovered by Smithson Tennant (England) in 1803. The origin of the name comes from the Latin word iris, meaning rainbow, because its salts are highly colored. It is heavy, brittle, white metal. Unreactive in air, water and acids. Attacked by fused NaOH. Metal ignites and burns readily. Iridium is found in gravel deposits with platinum. Used with osmium to tip gold pen points, to make crucible and special containers. Also to make alloys used for standard weights and measures and heat-resistant alloys. Also as hardening agent for platinum.
Citing this page:
Generalic, Eni. "Balance carried forward meaning." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table