amines → amini
Amines are compounds formally derived from ammonia by replacing one, two, or three hydrogen atoms by hydrocarbyl groups, and having the general structures RNH2 (primary amines), R2NH (secondary amines), R3N (tertiary amines). Examples are methylamine, CH3NH2; dimethylamine, CH3NHCH3; and trimethylamine, (CH3)3N.
amino acids → aminokiseline
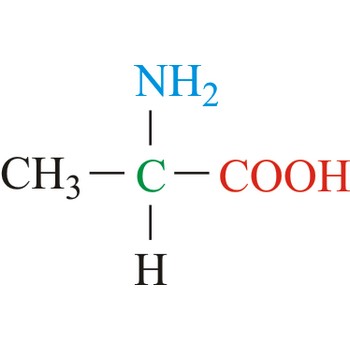
Amino acids are compounds containing both a carboxylic acid group (-COOH) and an amino group (-NH2 ). The most important are the α-amino acids, in which the -NH2 group in attached to the C atom adjacent to the -COOH group. In the β-amino acids, there is an intervening carbon atom.
primary amine → primarni amin
Primary amine is an amine on which there is only one alkyl group bonded, it is a weak base, and has a fish odour.
alanine → alanin
Alanine is hydrophobic amino acids with aliphatic side chain. It is the second simplest amino acid, but used the most in proteins. The nonpolar hydrophobic amino acids tend to cluster together within proteins, stabilizing protein structure by means of hydrophobic interactions. Alanine is a nonessential amino acid, meaning it can be manufactured by the human body, and does not need to be obtained directly through the diet.
- Abbreviations: Ala, A
- IUPAC name: 2-aminopropanoic acid
- Molecular formula: C3H7NO2
- Molecular weight: 89.09 g/mol
arginine → arginin
Arginine is an electrically charged amino acids with basic side chains. It is one of the least frequent amino acids. As a group the charged amino acids are important for making proteins soluble. These residues are generally located on the surface of the protein. Arginine is well designed to bind the phosphate anion, and is often found in the active centers of proteins that bind phosphorylated substrates. As a cation, arginine, as well as lysine, plays a role in maintaining the overall charge balance of a protein. Although arginine is considered an essential amino acid (it must be obtained through the diet), this is true only during the juvenile period in humans.
- Abbreviations: Arg, R
- IUPAC name: 2-amino-5-(diaminomethylideneamino)pentanoic acid
- Molecular formula: C6H14N4O2
- Molecular weight: 174.20 g/mol
conjugated protein → konjugirani protein
Conjugated proteins are proteins which have a prostetic group as a part of their structure which is bonded with one or more amino acids of the same protein.
nitrosoamine → nitrozoamin
Nitrosoamines are carcinogenic compounds that contain nitroso and amino-group in a single molecule.
asparagine → asparagin
Asparagine is neutral amino acids with polar side chains. The polar amino acids are an important class of amino acids since they provide many of the functional groups found in proteins. Asparagine is a common site for attachment of carbohydrates in glycoproteins. In general this is not very reactive residues. Asparagine is amide derivative of aspartic acid. Asparagine is not essential for humans, which means that it can be synthesized from central metabolic pathway intermediates and is not required in the diet.
- Abbreviations: Asn, N
- IUPAC name: 2,4-diamino-4-oxobutanoic acid
- Molecular formula: C4H8N2O3
- Molecular weight: 132.12 g/mol
Citing this page:
Generalic, Eni. "Amid." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table