electron configuration → elektronska konfiguracija
The electron configuration shows how many electrons there are in an atom or ion and their distribution along orbitals (see Table of electronic configuration of elements). Structure and all regularity in the periodic system depend upon electronic configuration of atoms of elements. Characteristics of elements mainly depend on electronic configuration of the outer shell. Refilling of the new electronic shell atoms of elements of similar electronic configuration emerge as well as in the previous shell, which adds up to periodicities of characteristics of elements.
fructose → fruktoza
Fructose (fruit sugar) is a ketohexose (a six-carbon ketonic sugar), which occurs in sweet fruits and honey. Glucose and fructose have the same molecular formula, C6H12O6, but have different structures. Pure, dry fructose is a very sweet, white, odorless, crystalline solid. Fructose is one of the sweetest of all sugars and is combined with glucose to make sucrose, or common table sugar. An older common name for fructose is levulose, after its levorotatory property of rotating plane polarized light to the left (in contrast to glucose which is dextrorotatory). The polysaccharide inulin is a polymer of fructose.
halogens → halogeni elementi
Halogens are the elements fluorine (F) chlorine (Cl), bromine (Br), iodine (I), and astatine (At). They are non-metals, and make up part of the 17 group in the periodic table. Compounds of these elements are called halogenides or halides.
The halogens all have a strong unpleasant odour and will burn flesh. They do not dissolve well in water. The five elements are strongly electronegative. They are oxidising agents, with fluorine being the strongest and astatine being the weakest. They react with most metals and many non-metals.
Halogens form molecules which consist of atoms covalently bonded. With increasing atomic weight there is a gradation in physical properties. For example: Fluorine is a pale green gas of low density. Chlorine is a greenish-yellow gas 1.892 times as dense as fluorine. Bromine is a deep reddish-brown liquid which is three times as dense as water. Iodine is a grayish-black crystalline solid with a metallic appearance. And astatine is a solid with properties which indicate that it is somewhat metallic in character.
Hesse’s law → Hessov zakon
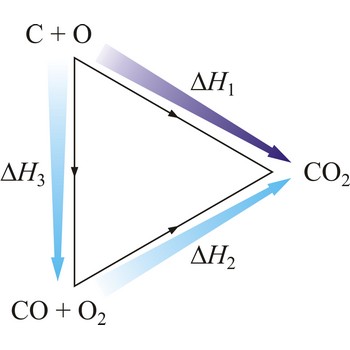
Hesse’s law says that reaction heat of some chemical change does not depend on the way in which the reaction is conducted, but only on starting and ending system state. Hesse’s law is also known as the law of constant heat summation. Hesse’s law is also known as the law of constant heat summation. The law was first put forward in 1840 by the Swiss-born Russian chemist Germain Henri Hess (1802-1850).
Hesse’s law can be used to obtain thermodynamic data that cannot be measured directly. For example, it is very difficult to control the oxidation of graphite to give pure CO. However, enthalpy for the oxidation of graphite to CO2 can easily be measured. So can the enthalpy of oxidation of CO to CO2. The application of Hess’s law enables us to estimate the enthalpy of formation of CO.
| C(s) + O2(g) →← CO2(g) | ΔrH1 = -393 kJ mol-1 |
| CO(g) + 1/2O2(g) →← CO2(g) | ΔrH2 = -283 kJ mol-1 |
| C(s) + 1/2O2(g) →← CO(g) | ΔrH3 = -110 kJ mol-1 |
The equation shows the standard enthalpy of formation of CO to be -110 kJ/mol.
homologous series → homologni niz
Series of compounds which have a common general formula and in which each member differs from the next member by a constant unit, which is the methylene group (-CH2-) is called the homologous series. Members of a homologous series are called homolog.
An example of the homologous series with some of their homologs are given below. Straight chain alkanes having general formula CnH2n+2
| Structure | Name |
|---|---|
| CH4 | methane |
| CH3-CH3 | ethane |
| CH3-CH2-CH3 | propane |
| CH3-CH2CH2CH3 | butane |
| CH3-(CH2)3-CH3 | pentane |
| CH3-(CH2)4-CH3 | hexane |
| CH3-(CH2)5-CH3 | heptane |
| CH3-(CH2)6-CH3 | octane |
| CH3-(CH2)7-CH3 | nonane |
| CH3-(CH2)8-CH3 | decane |
hydrosphere → hidrosfera
Hydrosphere (from the Greek for water sphere) is a discontinuous layer of water on, under, and over the Earth's surface. It includes all liquid and frozen surface waters, groundwater held in soil and rock, and atmospheric water vapour. Water continuously circulates between these reservoirs in what is called the hydrologic cycle, which is driven by energy from the Sun.
| Reservoir | V / 106 km3 | w / % |
|---|---|---|
| oceans | 1 370.0 | 97.25 |
| ice caps and glaciers | 29.0 | 2.05 |
| groundwater | 9.5 | 0.68 |
| lakes, rivers | 0.127 | 0.01 |
| soil moisture | 0.065 | 0.005 |
| atmosphere (as liquid equivalent of water vapour) | 0.013 | 0.001 |
| biosphere | 0.0006 | 0.00004 |
| TOTAL | 1 408.7 | 100 |
lanthanides → lantanoidi
Lanthanides (lanthanons, lanthanoids or rare-earth elements) are a series of fourteen elements in the periodic table, generally considered to range in proton number from cerium to lutetium inclusive. It was convenient to divide these elements into the cerium group or light earth: cerium (Ce), praseodymium (Pr), neodymium (Nd), promethium (Pm), samarium (Sm), europium (Eu); and the yttrium group or heavy earths: gadolinium (Gd), terbium (Tb), dysprosium (Dy), holmium (Ho), erbium (Er), thulium (Tm), ytterbium (Yb) i lutetium (Lu). The position of lanthanum is somewhat equivocal and, although not itself a lanthanide, it is often included with them for comparative purpose. The lanthanides are sometimes simply called the rare earths. Apart from unstable Pm, the lanthanides are actually not rare. Cerium is the 26. most abundant of all elements, 5 times as abundant as Pb. All are silvery very reactive metals.
logarithmic scale → logaritamska skala
Logarithmic scale is the one in which values of 1, 2, 3, 4, 5, in fact represents values of 1, 10, 100, 1 000, 10 000. Logarithmic scales are often used to simplify graphs and tables, where otherwise changes of data at the lower end of the scale would be difficult to distinguish (e.g. a graph axis which would normally have values from 1 - 1 000 000 is shown by values of 1 - 7). An example of a logarithmic scale is the pH scale.
mineral → mineral
Minerals are compounds in which metals can be found in nature. Metals in nature can appear as:
| autochthonous | Au, Cu, Pt, Ag, Pd, Hg, Ir |
| oxides | Fe, Al, Sn, Cr, Mn, W, Cu |
| sulphides | Cu, Pb, Zn, Ni, Ag, Co, Sb, Hg, Mo, Cd, Bi |
| carbonates | Fe, Zn, Cu, Mg, Mn, Pb |
| silicates | Ni, Cu, Zn, Mn |
| chlorides | Ag, Cu, Mg, Na, K |
| sulphates | Ca, Ba, Sr, Cu |
Moh’s scale → Mohsova skala
Mohs’ scale of mineral hardness characterises the scratch resistance of various minerals through the ability of a harder material to scratch a softer. It was created by the German mineralogist Friedrich Mohs (1773-1839). Mohs based the scale on the ten readily available minerals.
| Hardness | Mineral |
|---|---|
| 1 | talc (Mg3Si4O10(OH)2) |
| 2 | gypsum (CaSO4·2H2O) |
| 3 | calcite (CaCO3) |
| 4 | fluorite (CaF2) |
| 5 | apatite (Ca5(PO4)3(OH-,Cl-,F-)) |
| 6 | orthoclase feldspar (KAlSi3O8) |
| 7 | quartz (SiO2) |
| 8 | topaz (Al2SiO4(OH-,F-)2) |
| 9 | corundum (Al2O2) |
| 10 | diamond (C) |
Citing this page:
Generalic, Eni. "Panel tabi." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table