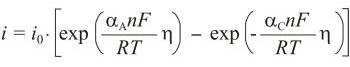antioxidant → antioksidans
Antioxidants are compounds that slow down oxidation processes that degrade foods, fuels, rubber, plastic, and other materials. Antioxidants like butylated hydroxyanisole (BHA), are added to food to prevent fats from becoming rancid and to minimize decomposition of vitamins and essential fatty acids; they work by scavenging destructive free radicals from the food.
atom marking → markiranje atoma
Atom marking is a process of infusing radioactive isotopes in live organism, with the purpose of revealing a way, diffusion, or a role of certain substance.
Bessemer, Henry → Bessemer, Henry
Sir Henry Bessemer (1813-1898) is the British engineer who first introduced a process for converting pig iron from a blast furnace into steel
air stripping → zračni striping
Air stripping is a process for the removal of volatile organic contaminants from groundwater. The groundwater flows downward inside a tower filled with materials (the packing) over a large surface area. Air is introduced at the bottom of the tower and is forced upward past the falling water. The volatiles evaporate from the water and are collected in air filters or released to the atmosphere.
battery → baterija
Battery a device that converts chemical energy to electrical energy. The process underlying the operation of a battery involves a chemical reaction in which electrons are transferred from one chemical species to another. This process is carried out in two half-reactions, one that involves the loss of electrons and one that involves their gain. The battery is an electrochemical cell divided in two half-cells, and reaction proceeds when these are connected together by an electrically conducting pathway. The passage of electrons from one half-cell to the other corresponds to an electric current. Each half-cell contains an electrode in contact with the reacting species. The electrode which passes electrons into the circuit when battery discharges is called anode and is negative terminal. The electrode which receives electrons is called cathode, and is the battery’s positive terminal. The electrical circuit is completed by an electrolyte, an electrically conducting substance placed between the two electrodes which carriers a flow of charge between them. In wet cells, the electrolyte is a liquid containing dissolved ions, whose motion generates an electrical current; in dry cells the electrolyte is basely solid, for example, a solid with mobile ions or porous solid saturated with an ionic solution.
biogas → bioplin
Biogas is a mixture of methane and carbon dioxide resulting from the anaerobic decomposition of such waste materials as domestic, industrial, and agricultural sewage. Methanogenic bacteria carry out the decomposition; these obligate anaerobes produce methane, the main component of biogas, which can be collected and used as an energy source for domestic processes, such as heating, cooking, and lighting.
blanching → blanširanje
1. Blanching is a heat treatment of foodstuffs to partially or completely inactivate the naturally occurring enzymes prior to freezing.
2. Blanching is a washing process for coins cleaning. The black surface layer of cupric oxide is removing by dipping the coins in hot dilute sulphuric acid (w(H2SO4) = 10 %).
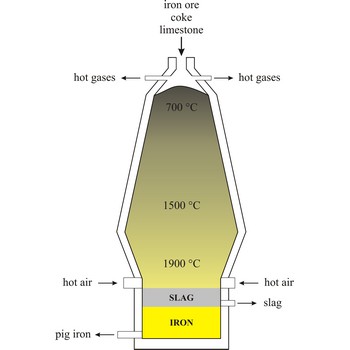
blast furnace → visoka peć
Blast furnace is a furnace for smelting of iron from iron oxide ores (hematite, Fe2O3 or magnetite, Fe3O4). Coke, limestone and iron ore are poured in the top, which would normally burn only on the surface. The hot air blast to the furnace burns the coke and maintains the very high temperatures that are needed to reduce the ore to iron. The reaction between air and the fuel generates carbon monoxide. This gas reduces the iron(III) oxide in the ore to iron.
Because the furnace temperature is in the region of 1500 °C, the metal is produced in a molten state and this runs down to the base of the furnace.
The production of iron in a blast furnace is a continuous process. The furnace is heated constantly and is re-charged with raw materials from the top while it is being tapped from the bottom. Iron making in the furnace usually continues for about ten years before the furnace linings have to be renewed.
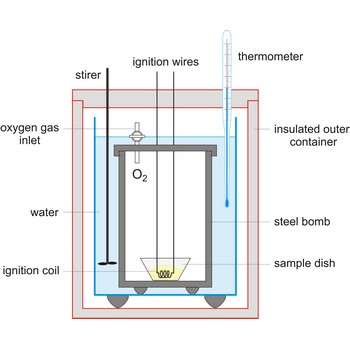
bomb calorimeter → kalorimetrijska bomba
Bomb calorimeter is a type of constant-volume calorimeter used in measuring the heat of combustion of samples which can be burned in oxygen. Four essential parts are required in any bomb calorimeter:
- a bomb or vessel in which the combustible charges can be burned,
- a bucket or container for holding the bomb in a measured quantity of water, together with a stirring mechanism,
- an insulating jacket to protect the bucket from transient thermal stresses during the combustion process, and
- a thermometer or other sensor for measuring temperature changes within the bucket.
Butler-Volmer equation → Butler-Volmerova jednadžba
Butler-Volmer equation is an activation controlled reaction, the one for which the rate of reaction is controlled solely by the rate of the electrochemical charge transfer process, which is in turn an activation-controlled process. This gives rise to kinetics that are described by the Butler-Volmer equation:
where io is exchange current density, η is overpotential (η = E - Eo), n is number of electrons, αA is anodic transfer coefficient, and αC is cathodic transfer coefficient
Citing this page:
Generalic, Eni. "Solvay%E2%80%99s process." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table