carbohydrate → ugljikohidrat
Carbohydrates (often called carbs for short) are polyhydroxy aldehydes or ketones, or substances that yield such compounds on hydrolysis. They are also known as saccharides, a term derived from the Latin word saccharum for sugar. Carbohydrates are the most abundant class of compounds in the biological world, making up more than 50 % of the dry weight of the Earth’s biomass. Every type of food we eat can have its energy traced back to a plant. Plants use carbon dioxide and water to make glucose, a simple sugar, in photosynthesis. Other carbohydrates such as cellulose and starch are made from the glucose. Light from the sun is absorbed by chlorophyll and this is converted to the energy necessary to biosynthesize carbohydrates
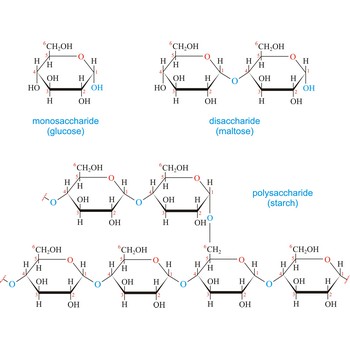
The term carbohydrate was applied originally to monosaccharides, in recognition of the fact that their empirical composition can be expressed as Cx(H2O)y. Later structural studies revealed that these compounds were not hydrates but the term carbohydrate persists.
Carbohydrates are generally classed as either simple or complex. Simple sugars, or monosaccharides, are carbohydrates that can’t be converted into smaller subunits by hydrolysis. Complex carbohydrates are made of two (disaccharides) or more (oligosaccharides, polysaccharides) simple sugars linked together by acetal (glycosidic) bonds and can be split into the former by hydrolysis.
contact procedure → kontaktni postupak
Contact procedure is an industrial procedure used for the production of sulphuric acid, where a dry and clean sulphur dioxide and air go over a catalyst made of vanadium pentoxide at 450 °C by which sulphur trioxide is gained, then we add concentrated sulphuric acid by which we obtain smoking sulphuric acid which is now diluted to sulphuric acid.
Dalton’s law → Daltonov zakon
Dalton’s law of partial pressure says that the total pressure eof gaseous mixture is equal to the sum of all gases partial pressures which make that mixture on the condition that they do not interact.
For example, if dry oxygen gas at 900 hPa is saturated with water vapor at 56 hPa, the pressure of the wet gas is 956 hPa.
desiccant dryers → sredstva za sušenje
Desiccant dryers (air dryers) use nonconsumable chemicals such as silica gel or active alumina, and can remove almost all the moisture from compressed air by adsorbing moisture on its surface. The method of regeneration, the process of removing adsorbed water from the desiccant, is the primary distinguishing feature among the various types of desiccant dryers. There are three ways to regenerate a desiccant: with air, internal or external heaters, or a heat pump.
desiccator → eksikator
Desiccator is a glass container with dry atmosphere due to the presence of some dehydrating agent. It is used for protecting the samples, reagents or precipitates from humidity. As dehydrating agent usually waterless calcium chloride (CaCl2) is used.
electrode potential → elektrodni potencijal
Electrode potential is defined as the potential of a cell consisting of the electrode in question acting as a cathode and the standard hydrogen electrode acting as an anode. Reduction always takes place at the cathode, and oxidation at the anode. According to the IUPAC convention, the term electrode potential is reserved exclusively to describe half-reactions written as reductions. The sign of the half-cell in question determines the sign of an electrode potential when it is coupled to a standard hydrogen electrode.
Electrode potential is defined by measuring the potential relative to a standard hydrogen half cell
The convention is to designate the cell so that the oxidised form is written first. For example
The e.m.f. of this cell is
By convention, at p(H2) = 101325 Pa and a(H+) = 1.00, the potential of the standard hydrogen electrode is 0.000 V at all temperatures. As a consequence of this definition, any potential developed in a galvanic cell consisting of a standard hydrogen electrode and some other electrode is attributed entirely to the other electrode
erbium → erbij
Erbium was discovered by Carl Gustaf Mosander (Sweden) in 1843. Named after Ytterby, a village in Sweden. It is soft, malleable, silvery metal. Reacts slowly with water. Dissolves in acids. Metal ignites and burns readily. Erbium is found with other heavier rare earths in xenotime and euxenite. Erbium oxide is used in ceramics to obtain a pink glaze. Also a few uses in the nuclear industry and as an alloying agent for other exotic metals.
gadolinium → gadolinij
Gadolinium was discovered by Jean de Marignac (France) in 1880. Named after the mineral gadolinite, named for J. Gadolin, a Finnish chemist and mineralogist. It is soft, ductile, silvery-white metal. Reacts slowly with water and oxygen. Dissolves in acids. Metal ignites and burns readily. Gadolinium is found with other rare earths in gadolinite and monazite sand. Used in steel alloying agents and the manufacture of electronic components.
half-cell → polučlanak
Half-cell is a part of galvanic cell in which oxidations or reduction of an element in contact with water or water solution one of its compounds.
holmium → holmij
Holmium was discovered by Per Theodore Cleve (Sweden) in 1879. The origin of the name comes from the Greek word Holmia meaning Stockholm. It is fairly soft, malleable, lustrous, silvery metal. Reacts slowly with oxygen and water. Dissolves in acids. Can react violently with air or halogens. Holmium occurs in gadolinite. Most often from monazite sand. It has very few practical applications; however, it has some unusual magnetic properties that offer some hope for future applications.
Citing this page:
Generalic, Eni. "Suhi članak." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table