wavelength → valna duljina
Wavelength is the distance between two neighbouring peaks of the electromagnetic wave.
wavefunction → valna funkcija
Wavefunction (Ψ) is a mathematical function that gives the amplitude of a wave as a function of position (and sometimes as a function of time and/or electron spin). Wavefunctions are used in chemistry to represent the behaviour of electrons bound in atoms or molecules.
wavenumber → valni broj
Wavenumber is the number of wave cycles per unit distance.
There are unfortunately two different definitions of the wavenumber.
Wavenumber, k, is most frequently defined as
with wavelength λ, phase velocity of wave vp, and angular frequency ω.
Less frequently it is defined simply as
One must be careful to note which definition is in use. Wavenumbers are used extensively in infrared spectroscopy, and usually have units of cm-1.
wave-particle duality → valno-čestična dualnost
Wave-particle duality is an observation that electrons, photons, and other very small entities behave like particles in some experiments and like waves in others.
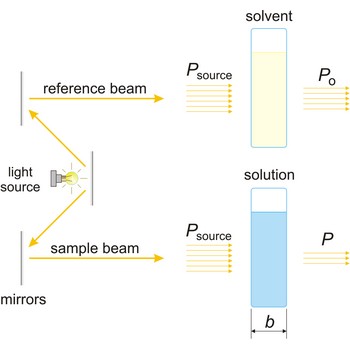
absorbance → apsorbancija
Absorbance (A) is a logarithm of the ratio of incident radiant power (Po) to transmitted radiant power (P) through a sample (excluding the effects on cell walls).
The absorption of light by a substance in a solution can be described mathematically by the Beer-Lambert law
where A is the absorbance at a given wavelength of light, ε is the molar absorbtivity or extinction coefficient (L mol-1 cm-1), unique to each molecule and varying with wavelength, b is the length of light path through the sample (cm), and c is the concentration of the compound in solution (mol L-1).
absorptance → apsorptancija
Absorptance (α) is the ratio of the radiant or luminous flux in a given spectral interval absorbed in a medium to that of the incident radiation. Also called absorption factor.
atomic absorption spectroscopy → atomska apsorpcijska spektroskopija
Atomic absorption spectroscopy (AAS) An analytical technique in which a sample is vapourised and the nonexcited atoms absorb electromagnetic radiation at characteristic wavelengths.
D-lines → D-linije
D-lines are two close lines in the yellow region of the spectrum of sodium, having wavelengths 589.6 nm (D1) and 589.0 nm (D2). They were labeled as feature D in the solar spectrum by German optician Joseph von Fraunhofer (1787-1826). As they a prominent and easily recognized they are used as a standard in spectroscopy.
diffraction grating → difrakcijska rešetka
Diffraction grating is a series of slits used to separate an incident wave into its component wavelengths by directionally separating their diffraction maxima.
Citing this page:
Generalic, Eni. "Valna duljina." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table