uranium → uranij
Uranium was discovered by Martin Heinrich Klaproth (Germany) in 1789. Named after the planet Uranus. It is silvery-white, dense, ductile, malleable, radioactive metal. Resists alkalis; tarnishes in air; attacked by steam and acids. Radiotoxic. Uranium occurs in many rocks, but in large amounts only in such minerals as pitchblende and carnotite. For many centuries it was used as a pigment for glass. Now it is used as a fuel in nuclear reactors and in bombs.
actinides → aktinoidi
Actinides (actinons or actinoids) are the fourteen elements from thorium to lawrencium inclusive, which follow actinium in the periodic table. The position of actinium is somewhat equivocal and, although not itself an actinide, it is often included with them for comparative purpose. The series includes the following elements: thorium (Th), protactinium (Pa), uranium (U), neptunium (Np), plutonium (Pu), amercium (Am), curium (Cm), berkelium (Bk), californium (Cf), einsteinium (Es), fermium (Fm), mendelevium (Md), nobelium (No) and lawrencium (Lr). Every known isotope of the actinide elements is radioactive. Traces of Pa, Np and Pu are consequently found, but only Th and U occur naturally to any useful extent.
actinium → aktinij
Actinium was discovered by André Debierne (France) in 1899. The origin of the name comes from the Greek word aktinos meaning ray. It is heavy, silvery-white, very radioactive metal. Reacts with water. Glows in the dark. Actinium is extremely rare, found in all uranium ores. Usually obtained by treating radium with neutrons in a reactor.
Curie → Curie
Maria Sklodowska-Curie (1867-1934) Polish-born French chemist who went to Paris in 1891. She married the physicist Pierre Curie (1859-1906) in 1985 and soon began work on seeking radioactive elements other than uranium in pitchblende (to account for its unexpectedly high radioactivity). By 1898 she had discovered radium and polonium although it took her years to purify them. In 1903 the Curies shared the Nobel Prize for physics with Henri Becquerel, who had discovered radioactivity.
einsteinium → einsteinij
Einsteinium was discovered by Albert Ghiorso (USA) in 1952. Named in honour of Albert Einstein (1879-1955). It is synthetic radioactive metal. Einsteinium was made by bombarding uranium with neutrons.
gamma radiation → gama-zračenje
Gamma radiation is electromagnetic radiation of extremely short wavelength. Gamma radiation ranges in energy from about 10-15 J to 10-10 J (10 keV to 10 MeV) (wavelength less than about 1 pm). Gamma rays are emitted by excited atomic nuclei during the process of passing to a lower excitation state.
Gamma rays are extremely penetrating and are absorbed by dense materials like lead and uranium. Exposure to gamma radiation may be lethal.
neptunium → neptunij
Neptunium was discovered by Edwin M. McMillan and P. H. Abelson (USA) in 1940. Named after the planet Neptune. It is rare, silvery radioactive metal. Resists alkalis; reacts with oxygen and acids. Attacked by steam. Radiotoxic. Neptunium was produced by bombarding uranium with slow neutrons.
transuranium element → trasuranski element
Transuranium elements are elements with an atomic number higher than 92 (uranium’s atomic number). Transuranium elements are unstable and occur in extremely low concentrations in nature. Most are made artificially.
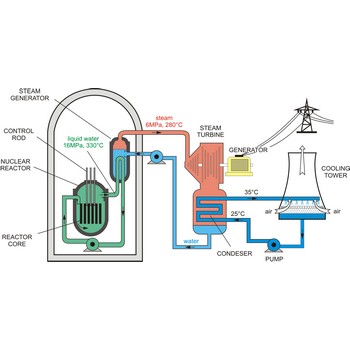
nuclear reactor → nuklearni reaktor
Nuclear reactor is an assembly of fissionable material (uranium-235 or plutonium-239) designed to produce a sustained and controllable chain reaction for the generation of electric power.
The essential components of a nuclear reactor are:
- The core, metal rods containing enough fissionable material to maintain a chain reaction at the necessary power level (as much as 50 t of uranium may be required).
- A source of neutrons to initiate the reaction (such as a mixture of polonium and beryllium)
- A moderator to reduce the energy of fast neutrons for more efficient fission (material such as graphite, beryllium, heavy water, and light water are used)
- A coolant to remove the fission-generated heat (water, sodium, helium, and nitrogen may be used)
- A control system such as rods of boron or cadmium that have high capture cross sections (to absorb neutrons)
- Adequate shielding, remote-control equipment, and appropriate instrumentation are essential for personnel safety and efficient operation.
plutonium → plutonij
Plutonium was discovered by Glenn T. Seaborg, Edwin M. McMillan, J. W. Kennedy and A. C. Wahl (USA) in 1940. Named after the planet Pluto. It is silvery-white, extremely radioactive artificially produced metal. Reacts with oxygen and acids; resists alkalis. Attacked by steam. Highly toxic. Plutonium is found rarely in some uranium ores. Made by bombarding uranium with neutrons. Used in bombs and reactors. Small quantities are used in thermo-electric generators.
Citing this page:
Generalic, Eni. "Uranij." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table