electrogravimetry → electrogravimetrija
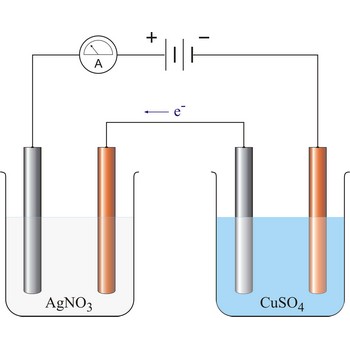
Electrogravimetry is an electroanalytical technique in which the substance to be determined (usually a metal) is deposited out on an electrode which is weighed before and after the experiment. The potential of the electrode must be carefully chosen to ensure that only the metal do be determined will deposit.
electrolysis → elektroliza
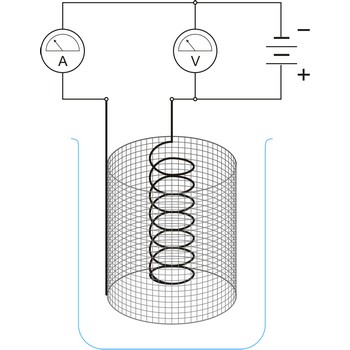
Electrolysis is the decomposition of a substance as a result of passing an electric current between two electrodes immersed in the sample.
electrophoresis → elektroforeza
Electrophoresis is a technique for the analysis and separation of colloids, based on the movement of charged colloidal particles in an electric field. The migration is toward electrodes of charge opposite to that of the particles. The rate of migration of the particles depends on the field, the charge on the particles, and on other factors, such as the size and shape of the particles.
Electrophoresis is important in the study of proteins. The acidity of the solution can be used to control the direction in which a protein moves upon electrophoresis.
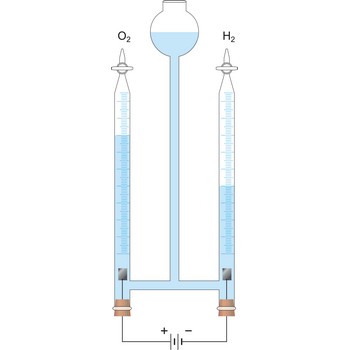
Faraday’s laws of electrolysis → Faradayevi zakoni elektrolize
Faraday’s laws of electrolysis are two laws found by British chemist and physicist Michael Faraday (1791-1867) in his experiments on electrolysis:
1. The quantity of matter extracted on the electrode is proportional to the quantity of charge (Q = I·t) which has flown in electrolysis time.
where z = number of electrons changed in reaction and F = Faraday’s constant which equals 96 487 C mol-1.
2. The masses of the elements liberated by the same quantity of electricity are directly proportional to their chemical equivalents.
96 487 C will discharge 1 mol Ag and 1/2 mol Cu. The relevant half reactions are:
filter paper → filtar papir
Filter paper is a quantitative paper used for filtering and made of pure cellulose treated with hydrochloric and hydrofluoric acid. This kind of paper burns out practically without any remains (less than 0.0001 g ashes). Different types of paper are marked with numbers; qualitative bears marking 595 or 597 and quantitative 589 or 590. Dependable upon precipitate character, different types of filter paper are used:
- black band (5891) - 100 mL of fluid flows through it in 20 s to 30 s. It is used for filtering of gelatinous precipitates.
- white band (5892) - 100 mL of fluid flows through it in 40 s to 60 s. It is used for coarse crystalline precipitates filtration.
- blue band (5893) - 100 mL of fluid flows through it in 200 s to 400 s. It is used for fine crystalline precipitates.
fuel cell → gorivi članak
Fuel cell is a device that converts chemical energy into electrical energy. It is different from a battery in that the energy conversion continues as long as fuel and oxidising agent are fed to the fuel cell; that is, in principle indefinitely. (A battery is manufactured with a limited amount of chemicals, and it is exhausted when all the chemicals have reacted.) It is a galvanic cell where spontaneous chemical reactions occur at the electrodes. The fuel is oxidised at the anode, and the oxidising agent (almost always oxygen or air) is reduced at the cathode. Presently, the most commonly used fuel is hydrogen. More conventional fuels (e.g., petrol or natural gas) must be converted (reformed) into hydrogen before they can be utilised in a fuel cell.
Some fuel cells employ an aqueous solution as electrolyte, that can be either acidic or basic (alkaline), or an ion-exchange membrane soaked in aqueous solution can act as the electrolyte. These fuel cells operate at relatively low temperatures (from room temperature to not much above the boiling point of water). Some fuel cells employ molten salts (especially carbonates) as electrolytes and have to operate at temperatures of several hundred degrees centigrade (Celsius). Others employ ionically conductive solids as electrolyte and must operate close to 1 000 °C.
Geiger counter → Geigerov brojač
Geiger counter (Geiger-Muller counter) is a device used to detect and measure ionising radiation. It consists of a tube containing a low-pressure gas (usually argon or neon with methane) and a cylindrical hollow cathode through the centre of which runs a fine-wire anode. A potential difference of about 1 000 V is maintained between the electrodes. An ionising particle or photon passing through a window into the tube will cause an ion to be produced and the high potential will accelerate it towards its appropriate electrode, causing an avalanche of further ionisations by collision. The consequent current pulses can be counted in electronic circuits or simply amplified to work a small loudspeaker in the instrument. It was first devised in 1908 by the German physicist Hans Geiger (1882-1945). Geiger and W. Muller produced an improved design in 1928.
gold → zlato
Gold has been known since ancient times. The origin of the name comes from the Latin word aurum meaning gold. It is soft, malleable, bright yellow metal. Unaffected by air, water, alkalis and most acids. Gold is found in veins in the crust, with copper ore and native. Used in electronics, jewellery and coins. It is a good reflector of infrared radiation, so a thin film of gold is applied to the glass of skyscrapers to reduce internal heating from sunlight.
galvanic celll → galvanski članak
Galvanic cell (voltaic cell) is a simple device with which chemical energy is converted into electrical energy. Galvanic cells consist of two separate compartments called half cells containing electrolyte solutions and electrodes that can be connected in a circuit. Two dissimilar metals (e.g., copper and zinc) are immersed in an electrolyte. If the metals are connected by an external circuit, one metal is reduced (i.e., gains electrons) while the other metal is oxidized (i.e., loses electrons).
In the example above, copper is reduced and zinc is oxidized. The difference in the oxidation potentials of the two metals provides the electric power of the cell.
A voltaic cell can be diagrammed using some simple symbols. In the diagram the electrodes are on the outer side of the diagram and a vertical line (|) is used to separate the electrode from the electrolyte solution found in the compartment. A double vertical line (||) is used to separate the cell compartments and is symbolic of the salt bridge. Usually in a diagram the species oxidized is written to the left of the double slash. Here is an example of the Daniell cell:
The names refer to the 18th-century Italian scientists Alessandro Volta (1745-1827) and Luigi Galvani (1737-1798).
hydrogen → vodik
Hydrogen was discovered by Sir Henry Cavendish (England) in 1766. The origin of the name comes from the Greek words hydro and genes meaning water and generate. It is colourless, odourless gas, burns and forms explosive mixtures in air. Reacts violently with oxidants. Hydrogen is the most abundant element in the universe. Commercial quantities of hydrogen are produced by reacting superheated steam with methane or carbon. In lab work from reaction of metals with acid solutions or electrolysis. Most hydrogen is used in the production of ammonia and in metal refining. Also used as fuel in rockets. Its two heavier isotopes (deuterium and tritium) used respectively for nuclear fusion.
Citing this page:
Generalic, Eni. "Srebro/srebrov klorid elektroda." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table