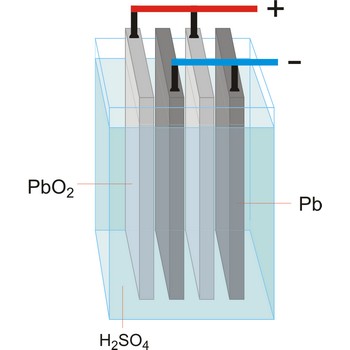
electrolytic cell → elektrolitska ćelija
Electrolytic cell is an electrochemical cell that converts electrical energy into chemical energy. The chemical reactions do not occur spontaneously at the electrodes when they are connected through an external circuit. The reaction must be forced by applying an external electric current. It is used to store electrical energy in chemical form (rechargeable battery). It is also used to decompose or produce (synthesise) new chemicals by the application of electrical power. This process is called electrolysis, e.g., water can be decomposed into hydrogen gas and oxygen gas. The free energy change of the overall cell reaction is positive.
Gibbs free energy → Gibbsova slobodna energija
Gibbs free energy (G) is an important function in chemical thermodynamics, defined by
where H is the enthalpy, S the entropy, and T the thermodynamic temperature. Gibbs free energy is the energy liberated or absorbed in a reversible process at constant pressure and constant temperature. Sometimes called Gibbs energy and, in older literature, simply free energy.
Changes in Gibbs free energy, ΔG, are useful in indicating the conditions under which a chemical reaction will occur. If ΔG is negative the reaction will proceed spontaneously to equilibrium. In equilibrium position ΔG = 0.
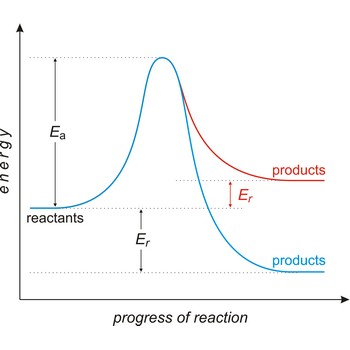
activation energy → energija aktivacije
Activation energy (Ea) is the energy that must be added to a system in order for a process to occur, even though the process may already be thermodynamically possible. In chemical kinetics, the activation energy is the height of the potential barrier separating the products and reactants. It determines the temperature dependence on the reaction rate.
acylaction reaction → reakcije aciliranja
Acylaction reaction involves the introduction of an acyl group (RCO-) into a compound. An alkyl halide is reacted with an alcohol or a carboxylic acid anhydride e.g.
The introduction of an acetyl group (CH3CO-) is acetylation, a process used for protecting -OH groups in organic synthesis.
adsorption → adsorpcija
Adsorption is a process in which molecules of gas, of dissolved substances in liquids, or of liquids adheres in an extremely thin layer to surfaces of solid bodies with which they are in contact.
antioxidant → antioksidans
Antioxidants are compounds that slow down oxidation processes that degrade foods, fuels, rubber, plastic, and other materials. Antioxidants like butylated hydroxyanisole (BHA), are added to food to prevent fats from becoming rancid and to minimize decomposition of vitamins and essential fatty acids; they work by scavenging destructive free radicals from the food.
atom marking → markiranje atoma
Atom marking is a process of infusing radioactive isotopes in live organism, with the purpose of revealing a way, diffusion, or a role of certain substance.
becquerel → bekerel
Becquerel (Bq) is the SI derived unit, with a special name, for radioactivity, equal to s-1. It describes a radioactivity of an amount of radionuclide decaying at the rate, on average, of one spontaneous nuclear transition per second. The unit was named after the French scientist Antoine Henri Becquerel (1852-1908) (disintegrations per unit time), equal to s-1.
air stripping → zračni striping
Air stripping is a process for the removal of volatile organic contaminants from groundwater. The groundwater flows downward inside a tower filled with materials (the packing) over a large surface area. Air is introduced at the bottom of the tower and is forced upward past the falling water. The volatiles evaporate from the water and are collected in air filters or released to the atmosphere.
Citing this page:
Generalic, Eni. "Spontani proces." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table