Results 1–10 of 10 for spektroskopija
atomic absorption spectroscopy → atomska apsorpcijska spektroskopija
Atomic absorption spectroscopy (AAS) An analytical technique in which a sample is vapourised and the nonexcited atoms absorb electromagnetic radiation at characteristic wavelengths.
atomic spectroscopy → atomska spektroskopija
Atomic spectroscopy is an expensive analytical method which uses absorption (AAS), emission (AES) and fluorescent (AFS) characteristics of the analyte.
infrared spectroscopy → infracrvena spektroskopija
Infrared (IR) spectroscopy is a technique used for determining the structure (and sometimes concentration) of molecules by observing how infrared radiation is absorbed by a sample.
spectroscopy → spektroskopija
Spectroscopy is the analysis of the lines of light emitted from excited atoms as the electrons drop back through their orbitals. These lines give the energy and distances of the electronic orbitals.
Bunsen burner → Bunsenov plamenik
Bunsen burner is a standard source of heat in the laboratory. German chemist Roberts Bunsen (1811-1899) improved the burner's design, which had been invented by Faraday, to aid his endeavors in spectroscopy. The Bunsen burner has a vertical metal tube through which a fine jet of fuel gas is directed. Air is drawn in through airholes near the base of the tube and the mixture is ignited and burns at the tube’s upper opening. The flow of this air is controlled by an adjustable collar on the side of the metal tube. When the whole is closed a yellow safety flame is displayed. Where as when the whole is open it displays a power dull blue flame with a faint blue outer flame with a vibrant blue core used u for combustion and hearting. The flame can reach temperatures of 1 500 °C.
Bunsen, Robert Wilhem → Bunsen, Robert Wilhem
Robert Wilhem Bunsen (1811-1899) is a German chemist who held professorships at Kassel, Marburg and Heidelberg. His early researches on organometallic compound of arsenic cost him an eye in an explosion. Bunsen's most important work was in developing several techniques used in separating, identifying, and measuring various chemical substances. He also improvement chemical battery for use in isolating quantities of pure metals - Bunsen battery.
The essential piece of laboratory equipment that has immortalized the name of Bunsen was not invented by him. Bunsen improved the burner's design, which had been invented by Faraday, to aid his endeavors in spectroscopy. Use of the Bunsen burner in conjunction with a glass prism led to the development of the spectroscope in collaboration with the German physicist Gustav Kirchoff and to the spectroscopic discovery of the elements rubidium (1860) and cesium (1861).
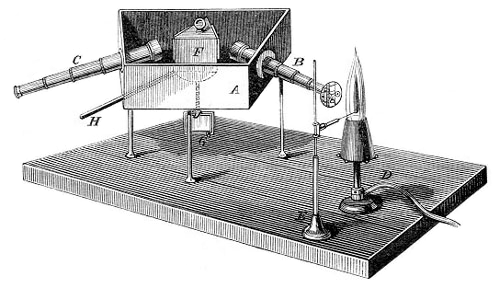
D-lines → D-linije
D-lines are two close lines in the yellow region of the spectrum of sodium, having wavelengths 589.6 nm (D1) and 589.0 nm (D2). They were labeled as feature D in the solar spectrum by German optician Joseph von Fraunhofer (1787-1826). As they a prominent and easily recognized they are used as a standard in spectroscopy.
nuclear magnetic resonance → nuklearna magnetska rezonancija
Nuclear magnetic resonance (NMR) is a type of radio-frequency spectroscopy based on the magnetic field generated by the spinning of electrically charged atomic nuclei. This nuclear magnetic field is caused to interact with a very large (1 T - 5 T) magnetic field of the instrument magnet. NMR techniques have been applied to studies of electron densities and chemical bonding and have become a fundamental research tool for structure determinations in organic chemistry.
wavenumber → valni broj
Wavenumber is the number of wave cycles per unit distance.
There are unfortunately two different definitions of the wavenumber.
Wavenumber, k, is most frequently defined as
with wavelength λ, phase velocity of wave vp, and angular frequency ω.
Less frequently it is defined simply as
One must be careful to note which definition is in use. Wavenumbers are used extensively in infrared spectroscopy, and usually have units of cm-1.
Citing this page:
Generalic, Eni. "Spektroskopija." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table

