electrochemical series → elektrokemijski niz
Electrochemical series is a series of chemical elements arranged in order of their standard electrode potentials. The hydrogen electrode
is taken as having zero electrode potential. An electrode potential is, by definition, a reduction potential.
Elements that have a greater tendency than hydrogen to lose electrons to their solution are taken as electropositive; those that gain electrons from their solution are below hydrogen in the series and are called electronegative.
The series shows the order in which metals replace one another from their salts; electropositive metals will replace hydrogen from acids.
electrode potential → elektrodni potencijal
Electrode potential is defined as the potential of a cell consisting of the electrode in question acting as a cathode and the standard hydrogen electrode acting as an anode. Reduction always takes place at the cathode, and oxidation at the anode. According to the IUPAC convention, the term electrode potential is reserved exclusively to describe half-reactions written as reductions. The sign of the half-cell in question determines the sign of an electrode potential when it is coupled to a standard hydrogen electrode.
Electrode potential is defined by measuring the potential relative to a standard hydrogen half cell
The convention is to designate the cell so that the oxidised form is written first. For example
The e.m.f. of this cell is
By convention, at p(H2) = 101325 Pa and a(H+) = 1.00, the potential of the standard hydrogen electrode is 0.000 V at all temperatures. As a consequence of this definition, any potential developed in a galvanic cell consisting of a standard hydrogen electrode and some other electrode is attributed entirely to the other electrode
lanthanides contraction → kontrakcija lantanoida
Lanthanides contraction is a reduction of metal and ion diameters from lanthanum to lutetium and it is caused by a core charge growth inside the same shell. Elements which in the periodic system of elements come after lanthanides have, because of lanthanides contraction, smaller diameter than they should have according to their position in the periodic system of elements.
oxidating agent → oksidans
Oxidating agent is a substance that receives electrons and oxidates other substances. Oxidating agent is always reduced in this reaction.
oxidation → oksidacija
The term oxidation originally meant a reaction in which oxygen combines chemically with another substance. More generally, oxidation is a part of a chemical reaction in which a reactant loses electrons (increases oxidation number). Simultaneous reduction of a different reactant must occur (redox reaction).
electroplating → galvaniziranje
Electroplating (also called electrodeposition) is the deposition of a metallic coating onto an object by putting a negative charge onto the object and immersing it into a solution which contains a salt of the metal to be deposited. The metallic ions of the salt carry a positive charge and are attracted to the part. When they reach it, the negatively charged part provides the electrons to reduce the positively charged ions to metallic form.
Typically, a brass or nickel object is coated with a layer of silver by making use of electrolysis of a silver solution, using the object to be coated as the cathode. The anode consist of pure silver, and the cathode is the object to be plated. The electrolyte is a mixure of silver nitrate with potassium cyanide. The reactions are:
The cyanide ensures a low concentration of silver ions, a condition for providing the best plating results.
Fehling’s test → Fehlingova otopina
Fehling’s test is a chemical test to detect reducing sugars and aldehydes in a solution, devised by the German chemist Hermann Christian von Fehling (1812-1885). Fehling’s solution consists of Fehling’s A (copper(II) sulphate solution) and Fehling’s B (sodium tartarate solution), equal amounts of which are added to the test solution. After boiling, a positive result is indicated by the formation of a brick-red precipitate of copper(I) oxide. Methanal, being a strong reducing agent, also produces copper metal; ketones do not react. The test is now rarely used, having been replaced by Benedict’s test.
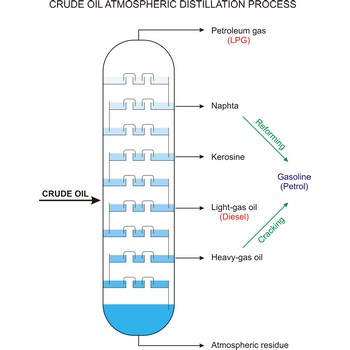
fractional distillation → frakcijska destilacija
Fractional distillation is a procedure in which liquids of close boiling points are separated. It is conducted in fraction or rectification columns in a way that vapour phase created by distillation is condensed and the condensate thus obtained is redistilled. The procedure is repeated several times. Vapour phase always contains more volatile component than the liquid phase, at top of the column vapour of clean volatile component gets out and at the bottom of the column liquid of nonvolatile component.
thermal pollution → toplinsko zagađenje
Thermal pollution is the increase in temperature of natural waters resulting from the discharge to these waters of hot effluents from industrial and power plants. The higher temperatures reduce the concentration of dissolved oxygen.
Citing this page:
Generalic, Eni. "Reakcije drugog reda." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table