water → voda
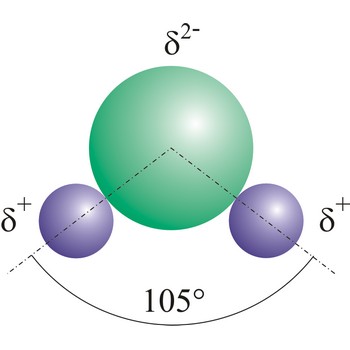
Water (H2O) (dihydrogen oxide) is a binary compound that occurs at room temperature as a clear colorless odorless tasteless liquid; freezes into ice below 0 °C and boils above 100 °C. Water is a chemical compound which is essential for living organisms and it is widely used as a solvent.
Wohler’s synthesis → Wohlerova sinteza
Wöhler’s synthesis is a synthesis of urea performed by the German chemist Friedrich Wöhler (1800-1882) in 1828. He discovered that urea (CO(NH2)2) was formed when a solution of ammonium isocyanate (NH4NCO) was evaporated. At the time it was believed that organic substances such as urea could only be made by living organisms, and its production from an inorganic compound was a notable discovery.
zinc → cink
Zinc was discovered by Andreas Marggraf (Germany) in 1746. The origin of the name comes from the German word zink. It is bluish-silver, ductile metal. Reacts with alkalis and acids. Tarnishes in air. Zinc is found in the minerals zinc blende (sphalerite) (ZnS), calamine, franklinite, smithsonite (ZnCO3), willemite and zincite (ZnO). Used to coat other metal (galvanizing) to protect them from rusting. Although some 90 % of the zinc is used for galvanizing steel. Zinc metal is used in the common dry-cell battery. Also used in alloys such as brass, bronze. Zinc compounds are also used in the manufacture of paints, cosmetics, plastics, electronic devices, and other products.
zwitterion → dipolarni ion
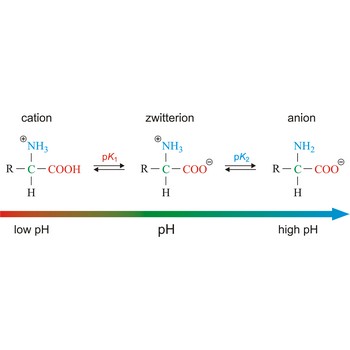
Zwitterion, also known as inner salt or dipolar ion, is an ion with a positive and a negative electrical charge at different locations within a molecule. As the molecule contains two opposite charges, it is electrically neutral. The term zwitterion is derived from the German word zwitter, meaning a hybrid, hermaphrodite. Zwitterions can be formed from compounds that contain both acid groups and base groups in their molecules (ampholytes).
All of the common amino acids found in proteins are ampholytes because they contain a carboxyl group (-COOH) that acts as an acid and an amino group (-NH2) that acts as a base. In the solid state, amino acids exist in the dipolar or zwitterion form. If acid is added to a solution containing the zwitterion, the carboxylate group captures a hydrogen (H+) ion, and the amino acid becomes positively charged. If base is added, ion removal of the H+ ion from the amino group of the zwitterion produces a negatively charged amino acid.
Dipeptide → Dipeptid
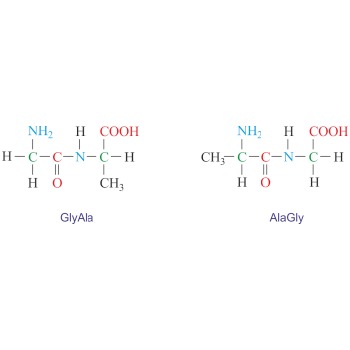
Dipeptide is an organic compound formed when two amino acids are joined by a peptide bond. Depending on which groups of amino acids are involved in the peptide bond four dipeptides can be formed from two different amino acids. For example, glycine (Gly) and alanine (Ala) can give two symmetrical dipeptides (GlyGly and AlaAla) and two unsymmetrical dipeptides (GlyAla and AlaGly). The naming is done by reading the sequence from the N-terminus to the C-terminus.
Citing this page:
Generalic, Eni. "Oxo compound." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table