normal boiling point → normalno vrelište
Normal boiling point is a temperature at which pressure of liquid vapour is 101 325 Pa.
boiling point → vrelište
Boiling point is the temperature at which the vapour pressure of a liquid is equal to the external pressure on the liquid. The standard boiling point is the temperature at which the vapour pressure of a liquid equals standard pressure (101 325 Pa).
boiling point elevation → povišenje vrelišta
Boiling point elevation is an elevation of the boiling point of a solvent is observed when substances are dissolved in it. The amount by which the boiling point is elevated is proportional to the number of molecules of solute and independent of their nature.
normal conditions → normalni uvjeti
Gas is under normal (or standard) conditions when: p0 = 105 Pa and T0 = 273.15 K (0 °C). IUPAC recommends that the former use of the pressure of 1 atm as standard pressure (equivalent to 101 325 Pa) should be discontinued. At these conditions, the molar volume of gas Vm0 is 0.022 711 m3 (22.711 L).
absolute temperature → apsolutna temperatura
Absolute temperature denoting a temperature measured on the absolute scale, a scale of temperature based on absolute zero as the lowest temperature.
heavy water → teška voda
Water molecules are composed of two hydrogen atoms and one oxygen atom (H2O). If the hydrogen atoms of a water molecule are replaced by deuterium atoms, the result is heavy water (D2O). Deuterium differs from hydrogen by having one neutron in the nucleus of the atom. There is approx. one part in 5000 D2O in normal water and it can be concentrated by electrolysis. Heavy water has a higher boiling point (101.4 °C) and melts at 3.6 °C. Heavy water is 20/18=1.11 times heavier than ordinary water.
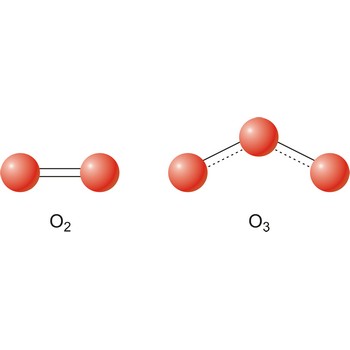
allotropic modification → alotropska modifikacija
Different substances of the same elementary system are called allotropes or allotropic modifications. In the case of oxygen, there are two allotropic modifications: "normal" dioxygen (O2) and trioxygen (O3) or ozone.
antifreeze → antifriz
Antifreeze is a substance added to the liquid (usually water) in the cooling systems of internal-combustion engines to lower its freezing point so that it does not solidify at sub-zero temperatures. The commonest antifreeze is ethane- 1.2-diol (ethylene glycol).
azeotrope → azeotropi
Azeotrope is a mixture of two liquids that boils at constant composition, i.e. the composition of the vapour is the same as that of the liquid. Azeotropes occur because of deviations in Raoult’s law leading to a maximum or minimum in the boiling point - composition diagram. The composition of an azeotrope depends on the pressure.
benzaldehyde → benzaldehid
Benzaldehyde (C6H5CHO) is a colourless volatile oily liquid. M.p. -26 °C, b.p. 178.1 °C. It is used in flavourings, perfumery and dyestuffs industry.
Citing this page:
Generalic, Eni. "Normalno vrelište." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table