Faraday cage → Faradayev kavez
Faraday cage is a container giving protection from electrical fields: an assembly of conducting material, for example, metal mesh or grid, placed around electrical equipment to protect it from external electrical fields. Faraday cages are named after the English scientist Michael Faraday (1791-1867).
fouling → obrastanje
1. Fouling is the deposition of insoluble materials, such as bacteria, colloids, oxides and water-borne debris, onto the surface of a reverse osmosis or ultrafiltration membrane. Fouling is associated with decreased flux rates and may also reduce the rejection rates of reverse osmosis membranes. Fouling also refers to the accumulation of normally inorganic deposits on heat exchanger tubing.
2. Fouling is an accumulation of marine organism deposits on a submerged metal surface.
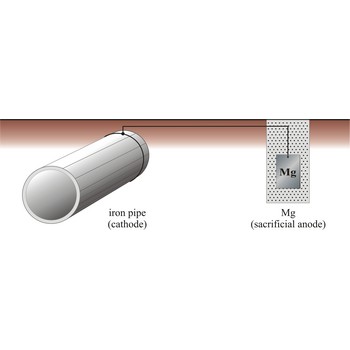
cathodic protection → katodna zaštita
Cathodic protection is a process in which a structural metal, such as iron, is protected from corrosion by connecting it to a metal that has a more negative reduction half-cell potential, which now corrodes instead of iron. There are two major variations of the cathodic method of corrosion protection. The first is called the impressed current method, and the other is called the sacrificial anode method.
chelate → kelat
Chelate is a compound characterized by the presence of bonds from two or more bonding sites within the same ligand to a central metal atom. For example, copper complexes with EDTA to form a chelate. Chelate complexes are more stable than complexes with the corresponding monodentate ligands.
complexometry → kompleksometrija
Complexometry is a volumetric analytic method which is based on titration of metal ion solutions with a substance that, combined with metal ions yields complex compounds, e.g. EDTA. The stoichiometric ratio of EDTA-metal in complexometric analyses is always 1:1, whatever the valency of the metal
corrosion → korozija
Corrosion is a harmful and undesirable construction material consumption by the chemical activity of its surroundings. Corrosion concept refers to metal and nonmetal construction materials, but it is usually used for metals, Corrosion of metal, according to the mechanism process, is divided into chemical (corrosion in nonelectrolytes) and electrochemical (corrosion in electrolytes).
Chemical corrosion appears by direct action of molecule of some element or compound on metal, thus directly creating corrosion products.
Electrochemical corrosion of metals occurs in electrolytes, so reduction of metal atom into free cation appears which by secondary processes gives molecules of compound which are considered a corrosion product.
immersion plating → galvaniziranje uranjanjem
Immersion plating involves depositing a metallic coating on a metal immersed in a liquid solution, without the aid of an external electric current. Also called dip plating.
lanthanides contraction → kontrakcija lantanoida
Lanthanides contraction is a reduction of metal and ion diameters from lanthanum to lutetium and it is caused by a core charge growth inside the same shell. Elements which in the periodic system of elements come after lanthanides have, because of lanthanides contraction, smaller diameter than they should have according to their position in the periodic system of elements.
ligand field theory → teorija ligandnog polja
Ligand field theory is a description of the structure of crystals containing a transition metal ion surrounded by nonmetallic ions (ligands). It is based on the construction of molecular orbitals involving the d-orbitals of the central metal ion and combinations of atomic orbitals of the ligands.
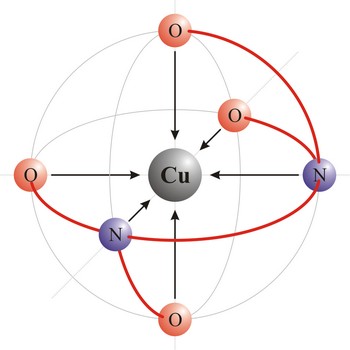
EDTA → EDTA
Ethyldiaminetetraacetic acid (C10H16N2O8) or shortened EDTA is a hexadentant ligand, and it forms chelates with both transition-metal ions and main-group ions. EDTA is used as a negative ion - EDTA4-. The diagram shows the structure of the ion with the important atoms picked out. The EDTA ion entirely wraps up a metal ion using all 6 of the positions. The co-ordination number is again 6 because of the 6 co-ordinate bonds being formed by the central metal ion.
EDTA is frequently used in soaps and detergents, because it forms a complexes with calcium and magnesium ions. These ions are in hard water and interfere with the cleaning action of soaps and detergents. EDTA is also used extensively as a stabilizing agent in the food industry and as an anticoagulant for stored blood in blood banks. EDTA is the most common reagent in complexometric titration.
Citing this page:
Generalic, Eni. "Metalni hidrid." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table