line spectrum → linijski spektar
Red-hot gases give line spectrum, i.e. is they emit electromagnetic rays of defined wavelengths. That kind of emission line of spectrum is characteristic of each chemical element.
electromagnetic radiation spectrum → spektar elektromagnetskog zračenja
Wavelengths of electromagnetic waves can be shown with the help of electromagnetic radiation spectrum. Electromagnetic radiation spectrum is divided into several areas from γ-radiation of very short wavelengths and great energy to radio waves with wavelengths up to 1 000 m. The human eye can only see a narrow part of the electromagnetic spectrum - visible radiation.
X-ray spectrum → spektar X-zraka
X-ray spectrum is a set of characteristic X-ray frequencies or wavelengths produced by a substance used as a target in an X-ray tube. Each element has a characteristic X-ray spectrum, and there is a strong correlation between atomic number and the frequencies of certain lines in the X-ray spectrum.
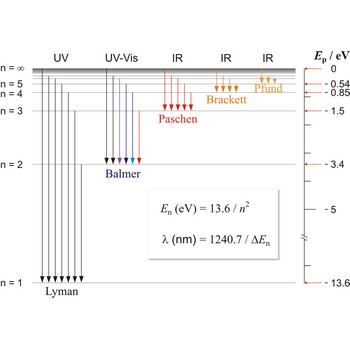
Balmer series → Balmerova serija
Balmer series, Balmer lines is a series of lines in the emission spectrum of hydrogen that involve transitions to the n=2 state from states with n>2.
Bohr atom → Bohrov atom
Bohr atom is a model of the atom that explains emission and absorption of radiation as transitions between stationary electronic states in which the electron orbits the nucleus at a definite distance. The Bohr model violates the Heisenberg uncertainty principle since it postulates definite paths and moment for electrons as they move around the nucleus. Modern theories usually use atomic orbitals to describe the behaviour of electrons in atoms.
chain → lanac
Chain is a linear combination of the same type of atom in a molecule. In straight chain molecules, the atoms are arranged in a line, with each atom in the chain linked to one preceding atom and one succeeding atom of the same type. A closed chain molecule is a chain where the atoms are linked in a ring structure.
infrared radiation → infracrveno zračenje
Infrared radiation is an electromagnetic radiation within the area from 1.0 μm to 300 μm, and is responsible for the transmission of radiant heat.
white light → bijela svjetlost
White light is a mixture of lights of all colours. If white light is passed through a glass prism or an optical lattice, it is separated into several colours (the visible light spectrum).
mass spectrometry → masena spectrometrija
Mass spectrometry is an analytical technique in which ions are separated according to the mass/charge (m/e) ratio and detected by a suitable detector.
In a mass spectrometer a sample is ionised and the positive ions produced are accelerated into a high-vacuum region containing electric and magnetic fields. These fields deflect and focus the ions onto a detector. A mass spectrum is thus obtained, consisting of a series of peaks of variable intensity to which m/e values can be assigned. Different molecules can be identified by their characteristic pattern of lines.
Citing this page:
Generalic, Eni. "Linijski spektar." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table