colligative properties → koligativna svojstva
Colligative properties are properties which affect a solvent based on the number of molecules of solute present such as melting point, boiling point and osmotic pressure.
chemical property → kemijsko svojstvo
Chemical property is a property observed when a substance undergoes a transformation into one or more new substances. Measurement of a chemical property involves a chemical change. For example, determining the flammability of petrol involves burning it, producing carbon dioxide and water.
extensive property → ekstenzivno svojstvo
Extensive property is a property that changes when the amount of matter in a sample changes. Examples are mass, volume, length, and charge.
intensive property → intenzivno svojstvo
intensive property is a property that does not change when the amount of sample changes. Examples are density, pressure, temperature, colour.
qualitative properties → kvalitativna svojstva
Qualitative properties are descriptional properties that cannot be defined by precise mathematical values, e.g. scent, taste and colour.
vapour pressure lowering → snižavanje tlaka pare
Vapour pressure is a colligative property of solutions. The vapour pressure of a solution is always lower than the vapour pressure of the pure solvent. Ratio of solution to pure solvent vapour pressures is approximately equal to the mole fraction of solvent in the solution.
absorbance → apsorbancija
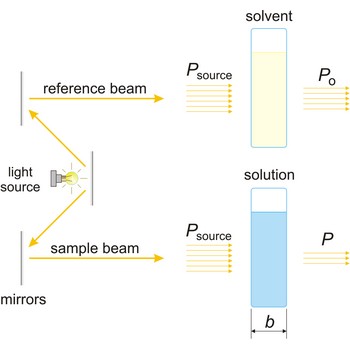
Absorbance (A) is a logarithm of the ratio of incident radiant power (Po) to transmitted radiant power (P) through a sample (excluding the effects on cell walls).
The absorption of light by a substance in a solution can be described mathematically by the Beer-Lambert law
where A is the absorbance at a given wavelength of light, ε is the molar absorbtivity or extinction coefficient (L mol-1 cm-1), unique to each molecule and varying with wavelength, b is the length of light path through the sample (cm), and c is the concentration of the compound in solution (mol L-1).
alicyclic → aliciklički spojevi
Alicyclic compounds are aliphatic compounds with a ring of atoms. They have CnH2n general formula (e.g. cyclohexane C6H12).
allotrope → alotrop
Allotropes are the elements which exist in two or more different forms in the same physical state. Allotropes generally differ in physical properties and may also differ in chemical activity.
Diamond, graphite and fullerenes are three allotropes of the element carbon. Graphite is a soft, black, slippery substance; by contrast, diamond is one of the hardest substances known. The different properties of the allotropes arise from their chemical structures. Diamonds typically crystallize in the cubic crystal system and consist of tetrahedrally bonded carbon atoms. Graphite crystallizes in the hexagonal system. In the fullerenes, the carbon atoms taking the form of a hollow sphere, ellipsoid, or tube.
In some cases, the allotropes are stable over a temperature range, with a definite transition point at which one changes into the other. For instance, tin has two allotropes: white (metallic) tin stable above 13.2 °C and grey (nonmetallic) tin stable below 13.2 °C.
The term allotropes may also be used to refer to the molecular forms of an element. Ozone is a chemically active triatomic allotrope of the element oxygen.
Citing this page:
Generalic, Eni. "Koligativno svojstvo." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table