activity coefficient → koeficijent aktiviteta
Activity coefficient (γ or f) is a fractional number which, when multiplied by the molar concentration of a substance in solution, yields the chemical activity. This term gives an idea of how much interaction exists between molecules at higher concentration.
In solutions of very low ionic strength, when m is less than 0.01, the Debye-Hückel limiting law can be used to calculate approximate activity coefficients
where γi = activity coefficient of the species i, zi = charge on the species i and μ = ionic strength of the solution.
absorption coefficient → apsorpcijski koeficijent
Absorption coefficient (a) is the relative decrease in the intensity of a collimated beam of electromagnetic radiation, as a result of absorption by a medium, during traversal of an infinitesimal layer of the medium, divided by the length traversed.
molar absorption coefficient → molarni apsorpcijski koeficijent
Molar absorption coefficient (ε) is the absorption coefficient divided by amount-of-substance concentration of the absorbing material in the sample solution (ε = a/c). The SI unit is m2mol-1. Also called extinction coefficient, but usually in units of dm3cm-1mol-1.
Joule-Thomson coefficient → Joule-Thompsonov koeficijent
Joule-Thomson coefficient (μ) is a parameter which describes the temperature change when a gas expands adiabatically through a nozzle from a high pressure to a low pressure region. It is defined by
where H is enthalpy.
stoichiometric coefficient → stehiometrijski koeficijent
Stoichiometric coefficient (ν) is the number appearing before the symbol for each compound in the equation for a chemical reaction. By convention, it is negative for reactants and positive for products.
Stoichiometric coefficients describe the stoichiometry of the chemical reaction.
In this equation, a, b, c and d are called as Stoichiometric coefficients of the A, B, C and D respectively.
activity → aktivitet
Activity (a) is a thermodynamic function used in place of concentration in equilibrium constants for reactions involving nonideal gases and solutions. For the species i activity is defined as
where ai is the activity of the species i, ci is its molar concentration, and fi is a dimensionless quantity called the activity coefficient.
Nernst’s electrode potential equation → Nernstova jednadžba za elektrodni potencijal
For general reaction of some redox system
dependence of electrode potential of redox system upon activity of oxidised and reduced form in solution is described in Nernst’s equation for electrode potential:
where E = to electrode potential of redox system
E° = standard electrode potential of redox system
R = universal gas constant
T = thermodymical temperature
F = Faraday’s constant
z = number of electrons exchanged in redox reaction
aO = activity of oxidised form
aR = activity of reduced form
n = stechiometrical coefficient of oxidised form
m = stechiometrical coefficient of reduced form
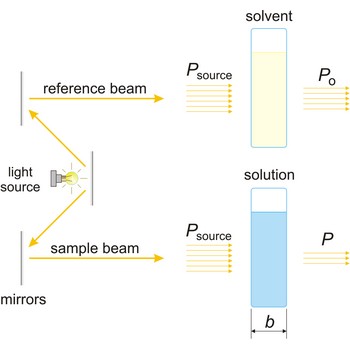
absorbance → apsorbancija
Absorbance (A) is a logarithm of the ratio of incident radiant power (Po) to transmitted radiant power (P) through a sample (excluding the effects on cell walls).
The absorption of light by a substance in a solution can be described mathematically by the Beer-Lambert law
where A is the absorbance at a given wavelength of light, ε is the molar absorbtivity or extinction coefficient (L mol-1 cm-1), unique to each molecule and varying with wavelength, b is the length of light path through the sample (cm), and c is the concentration of the compound in solution (mol L-1).
chemical equation equalization → izjednačavanje kemijske jednadžbe
Chemical equation equalization is determining values of stechiometric coefficients of reactants and products in a chemical equation in a way that the number of atoms of each element is equal before and after the reaction.
Fick’s law → Fickov zakon
Fick’s law is the statement that the flux J of a diffusing substance is proportional to the concentration gradient, i.e.,
where D is called the diffusion coefficient.
Citing this page:
Generalic, Eni. "Koeficijent aktiviteta." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table