anisotropic crystal → anizotropni kristal
Anisotropic crystal is a crystal that exhibits different properties when tested along axes in different directions.
anisotropy → anizotropija
Anisotropy is the property of molecules and materials to exhibit variations in physical properties along different molecular axes of the substance.
antioxidant → antioksidans
Antioxidants are compounds that slow down oxidation processes that degrade foods, fuels, rubber, plastic, and other materials. Antioxidants like butylated hydroxyanisole (BHA), are added to food to prevent fats from becoming rancid and to minimize decomposition of vitamins and essential fatty acids; they work by scavenging destructive free radicals from the food.
activated charcoal → aktivni ugljen
Activated charcoal or activated carbon is charcoal that has been activated for adsorption by steaming or by heating in a vacuum. Charcoal is obtained by burning wood, nutshells, coconut husks or other materials. Charcoal becomes activated by heating it with steam to approximately 1000 °C in the absence of oxygen.
The chemical nature of amorphous carbon, combined with a high surface area makes it an ideal medium for the adsorption of organic chemicals. A single gram of such material can have 400 m2 to 1 200 m2 square meters of surface area. Activated charcoal is widely used to decolorize liquids, recover solvents, and remove toxins from water and air.
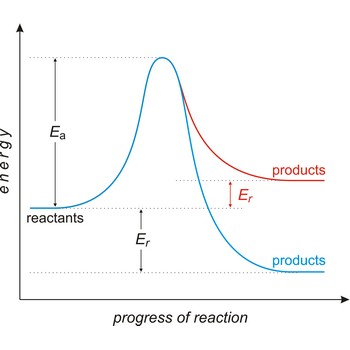
activation energy → energija aktivacije
Activation energy (Ea) is the energy that must be added to a system in order for a process to occur, even though the process may already be thermodynamically possible. In chemical kinetics, the activation energy is the height of the potential barrier separating the products and reactants. It determines the temperature dependence on the reaction rate.
adenosine triphosphate → adenozin trifosfat
Adenosine triphosphate (ATP) is nucleotide that is of fundamental importance as a carrier of chemical energy in all living organisms. It consists of adenin linked to D-ribose).
amount concentration → količinska koncentracija
Amount concentration (also called molar concentration and in older literature molarity) is the amount of a given substance in a stated unit of a mixture, solution, or ore. The common unit is mole per cubic decimetre (moldm−3) or mole per litre (molL-1) sometimes denoted by M.
The concentration of an atom, ion, or molecule in a solution may be symbolised by the use of square brackets, as [Ca2+].
antimony → antimon
Antimony has been known since ancient times. The origin of the name comes from the Latin word stibium meaning mineral stibnite. It is hard, brittle, silvery-white semimetal. Stable in dry air. Toxic by ingestion or inhalation. Antimony is found in stibnite (Sb2S3) and in valentinite (Sb2O3). It is alloyed with other metals to increase their hardness. Also in the manufacture of a few special types of semiconductor devices. Also in plastics and chemicals. A few kinds of over-the-counter cold and flu remedies use antimony compounds.
Citing this page:
Generalic, Eni. "Kemijsko svojstvo." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table