oxidation → oksidacija
The term oxidation originally meant a reaction in which oxygen combines chemically with another substance. More generally, oxidation is a part of a chemical reaction in which a reactant loses electrons (increases oxidation number). Simultaneous reduction of a different reactant must occur (redox reaction).
electrochemical series → elektrokemijski niz
Electrochemical series is a series of chemical elements arranged in order of their standard electrode potentials. The hydrogen electrode
is taken as having zero electrode potential. An electrode potential is, by definition, a reduction potential.
Elements that have a greater tendency than hydrogen to lose electrons to their solution are taken as electropositive; those that gain electrons from their solution are below hydrogen in the series and are called electronegative.
The series shows the order in which metals replace one another from their salts; electropositive metals will replace hydrogen from acids.
electrode of the first kind → elektroda prvog reda
Electrode of the first kind is a simple metal electrode immersed in a solution containing its own ion (e.g., silver immersed in a silver nitrate solution). The equilibrium potential of this electrode is a function of the concentration (more correctly of activity) of the cation of the electrode metal in the solution (see Nernst’s electrode potential equation).
electrode of the second kind → elektroda drugog reda
Electrodes of the second kind are metal electrodes assembly with the equilibrium potential being a function of the concentration of an anion in the solution. Typical examples are the silver/silver-chloride electrode and the calomel electrode. The potential of the metal is controlled by the concentration of its cation in the solution, but this, in turn, is controlled by the anion concentration in the solution through the solubility product of the slightly soluble metal salt. Contrast with electrode of the first kind and electrode of the third kind.
electrode of the third kind → elektroda trećeg reda
Electrode of the third kind is a metal electrode assembly with the equilibrium potential being a function of the concentration of a cation, other than the cation of the electrode metal, in the solution. The assembly consists of a metal in contact with two slightly soluble salts (one containing the cation of the solid metal, the other the cation to be determined, with both salts having a common anion) immersed in a solution containing a salt of the second metal (e.g., zinc metal--zinc oxalate--calcium oxalate--calcium salt solution). The potential of the metal is controlled by the concentration of its cation in the solution, but this is controlled by the anion concentration in the solution through the solubility product of the slightly soluble metal salt, which, in turn is controlled by the concentration of the cation of the second slightly soluble salt. These electrodes are very sluggish and unstable due to a series of equilibria to be established to produce a stable potential.
permeability → permeabilnost
Permeability (Latin permeare, to pass through) is a passage or diffusion of a gas, vapour, liquid, or solid through a material without physically or chemically affecting it.
polymorphic transition → polimorfni prijelaz
Polymorphic transition is a reversible transition of a solid crystalline phase at a certain temperature and pressure to another phase of the same chemical composition with a different crystal structure. For examples, the transitions of quartz (SiO2) at 1 143 K to tridymite, and at 1 743 K to cristobalite.
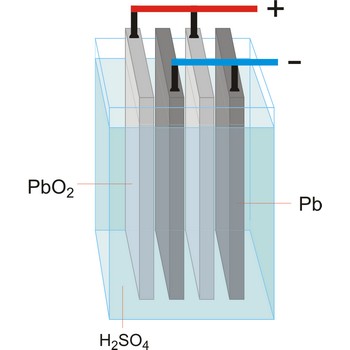
electrolytic cell → elektrolitska ćelija
Electrolytic cell is an electrochemical cell that converts electrical energy into chemical energy. The chemical reactions do not occur spontaneously at the electrodes when they are connected through an external circuit. The reaction must be forced by applying an external electric current. It is used to store electrical energy in chemical form (rechargeable battery). It is also used to decompose or produce (synthesise) new chemicals by the application of electrical power. This process is called electrolysis, e.g., water can be decomposed into hydrogen gas and oxygen gas. The free energy change of the overall cell reaction is positive.
Citing this page:
Generalic, Eni. "Kemijska ravnoteža." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table