brass → mjed
Brasses are alloys of copper and zinc (generally 5 % to 40 %). Brass has been known to man since prehistoric times, long before zinc itself was discovered. It was produced by melting copper together with calamine, a zinc ore. Its ductility reaches a maximum with about 30 % zinc and its tensile strength with 45 % although this property varies greatly with the mechanical and heat treatment of the alloy. Typical applications included gears, plumbing ware fittings, adapters, valves and screw machine products. The French horn is a valved brass wind instrument.
Brass may contain small amounts of other alloying elements, such as aluminum, lead, tin, or nickel. Lead can be added as an alloying element resulting in a brass that can be rapidly machined and produces minimal tool wear. Additions of aluminium, iron and manganese to brass improve strength, whilst silicon additions improve wear resistance. Brass containing tin (< 2 % ) is less liable to corrosion in seawater; it is sometimes called naval brass and is used in naval construction.

bromine → brom
Bromine was discovered by Antoine J. Balard (France) in 1826. The origin of the name comes from the Greek word bromos meaning stench. It is reddish-brown liquid with suffocating, irritating fumes. Gives off poisonous vapour. Causes severe burns. Oxidizer. Bromine occurs in compounds in sea water. It was once used in large quantities to make a compound that removed lead compound build up in engines burning leaded gasoline. Now it is primarily used in dyes, disinfectants and photographic chemicals.
bronze → bronca
Bronze is an alloy made primarily of copper and tin. It may contain as much as 25 % tin. Bronzes with 10 % or more tin are harder, stronger, and resistant to corrosion. As bronze weathers, a brown or green film forms on the surface. This film inhibits corrosion. Silicon or aluminium is often added to bronze to improve resistance to corrosion. Phosphorus, lead, zinc, and other metals may be added for special purposes. The alloy is hard and easily cast and is extensively used in bearings, valves and other machine parts.
Bronze was one of the first alloys developed by ancient metal workers. The Bronze Age occurred in Europe around 2200 to 700 BC. Bronze was used for weapons such as spearheads, swords, and knives. Since ancient times, bronze has been the most popular metal for casting statues and other art objects.

The term bronze has been adopted commercially for many copper-rich alloys that contain little or no tin but are similar in colour to bronze, including aluminium bronze, manganese bronze, and silicon bronze. Aluminium bronze is used to make tools and, because it will not spark when struck. Manganese bronze is actually a brass that contains manganese. It is often used to make ship propellers because it is strong and resists corrosion by sea water.
cadmium → kadmij
Cadmium was discovered by Friedrich Strohmeyer (Germany) in 1817. The origin of the name comes from the Latin word cadmia meaning calamine (zinc carbonate, ZnCO3), or from the Greek word kadmeia with the same meaning. It is soft, malleable, blue-white metal. Tarnishes in air, soluble in acids, insoluble in alkalis. Boiling cadmium gives off a weird, yellow-colored vapour that is poisonous. Cadmium can cause a variety of health problems, including kidney failure and high blood pressure. Cadmium is obtained as a by product of zinc refining. The mayor use of cadmium is in electroplating of steel to protect it from corrosion. Also used to make nickel-cadmium batteries. The ability of cadmium to adsorb neutrons has made it of great importance in the design of nuclear reactors. Its compounds are found in paint pigments and a wide variety of intense colours.
caesium → cezij
Caesium was discovered by Robert Bunsen and Gustav Kirchhoff (Germany) in 1860. The origin of the name comes from the Latin word caesius meaning sky blue or heavenly blue. It is very soft, light grey, ductile metal. Reacts readily with oxygen. Reacts explosively with water. Caesium is found in pollucite [(Cs4Al4Si9O26)·H2O] and as trace in lepidolite. Used as a ’getter’ to remove air traces in vacuum and cathode-ray tubes. Also used in producing photoelectric devices and atomic clocks. Since it ionises readily, it is used as an ion rocket motor propellant.
calcium → kalcij
Calcium was discovered by Sir Humphry Davy (England) in 1808. The origin of the name comes from the Latin word calix meaning lime. It is fairly hard, silvery-white metal. Exposed surfaces form oxides and nitrides. Reacts with water and oxygen. Occurs only in compounds. Calcium is obtained from minerals like chalk, limestone and marble. Pure metal is produced by replacing the calcium in lime (CaCO3) with aluminium in hot, low pressure retorts. Used by many forms of life to make shells and bones. Virtually no use for the pure metal, however two of its compounds are, lime (CaO) and gypsum (CaSO4), are in great demand by a number of industries.
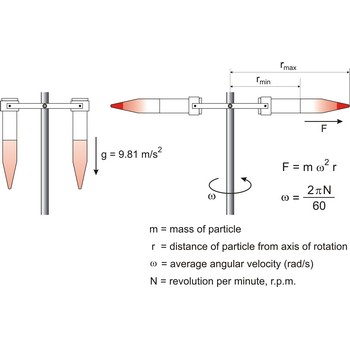
centrifugation → centrifugiranje
Centrifugation is sedimentation of particles under the influence of the centrifugal force and it is used for separation of superfine suspensions. At centrifuging forces up to 10 000 times greater than gravity force are used, and at ultracentrifuge up to 600 000 times as great.
Citing this page:
Generalic, Eni. "Grej." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table