mass spectrometry → masena spectrometrija
Mass spectrometry is an analytical technique in which ions are separated according to the mass/charge (m/e) ratio and detected by a suitable detector.
In a mass spectrometer a sample is ionised and the positive ions produced are accelerated into a high-vacuum region containing electric and magnetic fields. These fields deflect and focus the ions onto a detector. A mass spectrum is thus obtained, consisting of a series of peaks of variable intensity to which m/e values can be assigned. Different molecules can be identified by their characteristic pattern of lines.
plasma → plazma
Plasma is a highly ionised gas in which the charge of the electrons is balanced by the charge of the positive ions, so that the system as a whole is electrically neutral. Plasmas are created by exposing gases at low pressure to an electric or electromagnetic field. In semiconductor processing, plasmas are used for etching and thin film deposition (the excited state of the gas makes it very reactive). In everyday life plasmas are used to give light in fluorescent light bulbs, neon lamps, and blue insect traps.
solar cell → sunčeva ćelija
Solar cell, or photovoltaic cell, is a device that captures sunlight and transforms it directly to electricity. All solar cells make use of photovoltaic effect, so often they are called photovoltaic cells. Almost all solar cells are built from solid-state semiconducting materials, and in the vast majority of these the semiconductor is silicon.
The photovoltaic effect involves the generation of mobile charge carriers-electrons and positively charged holes-by the absorption of a photon of light. This pair of charge carriers is produced when an electron in the highest filled electronic band of a semiconductor (the valence band) absorbs a photon of sufficient energy to promote it into the empty energy band (the conduction band). The excitation process can be induced only by a photon with an energy corresponding to the width of the energy gap that separates the valence and the conduction band. The creation of an electron-hole pair can be converted into the generation of an electrical current in a semiconductor junction device, wherein a layer of semiconducting material lies back to back with a layer of either a different semiconductor or a metal. In most photovoltaic cells, the junction is p-n junction, in which p-doped and n-doped semiconductors are married together. At the interface of the two, the predominance of positively charged carriers (holes) in the p-doped material and of negatively charged carriers (electrons) in the n-doped material sets up an electric field, which falls off to either side of the junction across a space-charge region. When absorption of a photon in this region generates an electron-hole pair, these charge carriers are driven in opposite directions by the electric field, i.e. away from the interface and toward the top and bottom of the two-layer structure, where metal electrodes on these faces collect the current. The electrode on the top layer (through which light is absorbed) is divided into strips so as not to obscure the semiconducting layers below. In most widely used commercial solar cells, the p-doped and n-doped semiconductive layers are formed within a monolithic piece of crystalline silicon. Silicon is able to absorb sunlight at those wavelengths at which it is most intense-from the near-infrared region (wavelengths of around 1200 nm) to the violet (around 350 nm).
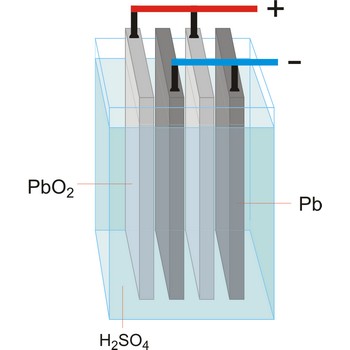
accumulator → akumulator
Accumulator (secondary cell, storage battery) is a type of voltaic cell or battery that can be recharged by passing current through it from an external D.C. supply. The charging current reverses the chemical reactions in the cell. The common types are the lead-acid accumulator and the nickel-cadmium cell.
Acheson process → Achesonov proces
Acheson process is an industrial process to synthesize graphite and silicon carbide (carborundum), named after its inventor the American chemist Edward Goodrich Acheson (1856-1931). In this process, a solid-state reaction between pure silica sand (SiO2) and petroleum coke (C) at very high temperature (more than 2500 °C) leads to the formation of silicon carbide under the general reaction:
While studying the effects of high temperature on carborundum, Acheson had found that silicon vaporizes at about 4150 °C, leaving behind graphitic carbon.
aluminium → aluminij
Aluminium was discovered by Friedrich Wöhler (Germany) in 1827. The origin of the name comes from the Latin word alumen meaning alum. It is soft, lightweight, silvery-white metal. Exposed surfaces quickly form protective oxide coating. Metal reacts violently with oxidants. Third most abundant element in the earth’s crust. Aluminium is the most abundant metal to be found in the earth’s crust, but is never found free in nature. Aluminium is obtained by electrolysis from bauxite. Used for many purposes from airplanes to beverage cans. Too soft in its pure form so less than 1 % of silicon or iron is added, which hardens and strengthens it.
barrier film → barijerni film
Barrier film is a thin, continuous, non-porous, electrically insulating film on metal surfaces (usually comprised of oxides).
biogas → bioplin
Biogas is a mixture of methane and carbon dioxide resulting from the anaerobic decomposition of such waste materials as domestic, industrial, and agricultural sewage. Methanogenic bacteria carry out the decomposition; these obligate anaerobes produce methane, the main component of biogas, which can be collected and used as an energy source for domestic processes, such as heating, cooking, and lighting.
conductivity → vodljivost
Conductivity is a measure of the ability of a material to conduct electric current.
Citing this page:
Generalic, Eni. "Električno polje." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table