Snell’s law → Snellov zakon
When a light ray comes on a boundary between two transparent media, it will be partly reflected and partly refracted. Both rays, reflected and refracted ray, lay in the plane of incidence. The angle of reflection is equal to the angle of incidence. The angle of refraction (Θ2) is related to the angle of incidence (Θ1) via Snell’s law:
where n1 and n2 are dimensionless constants - indexes of refraction of the two media.
sodium → natrij
Sodium was discovered by Sir Humphry Davy (England) in 1807. The origin of the name comes from the Latin word natrium meaning sodium carbonate. It is soft silvery-white metal. Fresh surfaces oxidize rapidly. Reacts vigorously, even violently with water. Reacts with water to give off flammable gas. Burns in air with a brilliant white flame. Sodium is obtained by electrolysis of melted sodium chloride (salt), borax and cryolite. Metallic sodium is vital in the manufacture of organic compounds. Sodium chloride (NaCl) is table salt. Liquid sodium is used to cool nuclear reactors.
solar cell → sunčeva ćelija
Solar cell, or photovoltaic cell, is a device that captures sunlight and transforms it directly to electricity. All solar cells make use of photovoltaic effect, so often they are called photovoltaic cells. Almost all solar cells are built from solid-state semiconducting materials, and in the vast majority of these the semiconductor is silicon.
The photovoltaic effect involves the generation of mobile charge carriers-electrons and positively charged holes-by the absorption of a photon of light. This pair of charge carriers is produced when an electron in the highest filled electronic band of a semiconductor (the valence band) absorbs a photon of sufficient energy to promote it into the empty energy band (the conduction band). The excitation process can be induced only by a photon with an energy corresponding to the width of the energy gap that separates the valence and the conduction band. The creation of an electron-hole pair can be converted into the generation of an electrical current in a semiconductor junction device, wherein a layer of semiconducting material lies back to back with a layer of either a different semiconductor or a metal. In most photovoltaic cells, the junction is p-n junction, in which p-doped and n-doped semiconductors are married together. At the interface of the two, the predominance of positively charged carriers (holes) in the p-doped material and of negatively charged carriers (electrons) in the n-doped material sets up an electric field, which falls off to either side of the junction across a space-charge region. When absorption of a photon in this region generates an electron-hole pair, these charge carriers are driven in opposite directions by the electric field, i.e. away from the interface and toward the top and bottom of the two-layer structure, where metal electrodes on these faces collect the current. The electrode on the top layer (through which light is absorbed) is divided into strips so as not to obscure the semiconducting layers below. In most widely used commercial solar cells, the p-doped and n-doped semiconductive layers are formed within a monolithic piece of crystalline silicon. Silicon is able to absorb sunlight at those wavelengths at which it is most intense-from the near-infrared region (wavelengths of around 1200 nm) to the violet (around 350 nm).
spectrophotometer → spektrofotometar
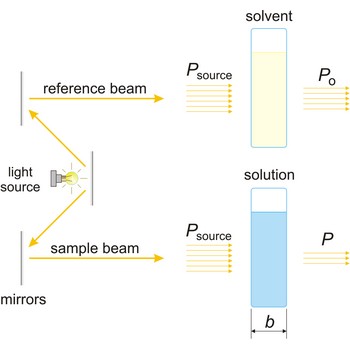
Spectrophotometer is an instrument for measuring the amount of light absorbed by a sample.
The absorption of light by a substance in a solution can be described mathematically by the Beer-Lambert law
where A is the absorbance at a given wavelength of light, ε is the molar absorbtivity or extinction coefficient (L mol-1 cm-1), unique to each molecule and varying with wavelength, b is the length of light path through the sample (cm), and c is the concentration of the compound in solution (mol L-1).
strontium → stroncij
Strontium was discovered by Sir Humphry Davy (England) in 1808. Named after the village of Strontian in Scotland. It is soft, malleable, silvery-yellow metal. Combustible in air, will react with water. Exposed surfaces form protective oxide film. Metal ignites and burns readily. Strontium is found in minerals celestite and strontianite. Used in flares and fireworks for crimson colour. Strontium-90 is a long lived highly radioactive fallout product of atomic-bomb explosions.
sucrose → saharoza
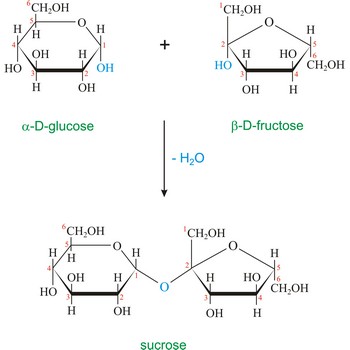
Sucrose (saccharose), or ordinary table sugar, is a disaccharide in which α-D-glucopyranose and β-D-fructofuranose are joined at their anomeric carbons by a glycosidic bond. There are no hemiacetals remaining in the sucrose and therefore sucrose is not a reducing sugar and does not exhibit mutarotation. Sugar is a white crystalline sweet compound found in many plants and extracted from sugar cane and sugar beet. It is used as a sweetening agent in food and drinks. If heated to 200 °C, sucrose becomes caramel. When sucrose is hydrolyzed it forms an equimolar mixture of glucose and fructose. This mixture of monosaccharides is called invert sugar. Honeybees have enzymes called invertases that catalyze the hydrolysis of sucrose. Honey, in fact, is primarily a mixture of glucose, fructose, and sucrose.
tellurium → telurij
Tellurium was discovered by Franz Joseph Muller von Reichstein (Romania) in 1782. The origin of the name comes from the Latin word tellus meaning earth. It is silvery-white, brittle semi-metal. Unreactive with water or HCl; dissolves in HNO3; burns in air or oxygen. Tellurium is obtained as a by-product of copper and lead refining. Used to improve the machining quality of copper and stainless steel products and to colour glass and ceramics. Also in thermoelectric devices. Some is used in the rubber industry and it is a basic ingredient in manufacturing blasting caps.
temporary hardness → prolazna tvrdoća
Temporary Hardness is due to the bicarbonate ion, HCO3-, being present in the water. It can be removed by water reboiling, whereby white solid emerges calcium carbonate that is limescale.
tin → kositar
Tin has been known since ancient times. The origin of the name comes from the Latin word stannum meaning tin. It is silvery-white, soft, malleable and ductile metal. Exposed surfaces form oxide film. Resists oxygen and water. Dissolves in acids and bases. Organic tin compounds may be highly toxic. Tin is principally found in the ore cassiterite (SnO2) and stannine (Cu2FeSnS4). Used as a coating for steel cans since it is non-toxic and non-corrosive. Also in solder (33 %Sn:67 %Pb), bronze (20 %Sn:80 %Cu) and pewter. Stannous fluoride (SnF2), a compound of tin and fluorine is used in some toothpaste.
tungsten → volfram
Tungsten was discovered by Fausto and Juan Jose de Elhuyar (Spain) in 1783. Named after the tungsten mineral wolframite. It is hard, steel-grey to white metal. Highest melting point of all metals. Resists oxygen, acids and alkalis. Tungsten occurs in the minerals scheelite (CaWO4) and wolframite [(Fe,Mn)WO4]. Made into filaments for vacuum tubes and electric lights. Also as contact points in cars. Tungsten carbide is extremely hard and is used for making cutting tools and abrasives.
Citing this page:
Generalic, Eni. "Bijela svjetlost." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table