U-tube manometer → U-manometar
U-tube manometer contains water or mercury in a U-shaped tube, and is usually used to measure gas pressure. One end of the U tube is exposed to the unknown pressure field (P) and the other end is connected to a reference pressure source (usually atmospheric pressure) (Pref), shown in the schematic below.
If fluid C is the atmosphere, fluid B is the liquid in the U tube (e.g. water or mercury), and fluid A is a gas, then we can assume that ρB >> ρA, ρC. The pressure contributed by the weight of gas within the U tube can therefore be neglected. The gage pressure of the gas can be approximated by,
absolute temperature → apsolutna temperatura
Absolute temperature denoting a temperature measured on the absolute scale, a scale of temperature based on absolute zero as the lowest temperature.
aerosol → aerosol
Aerosols are colloidal dispersions of liquid or solid particles in a gas, as in a mist or smoke. The commonly used aerosol sprays contain an inert propellant liquefied under pressure. The pressure of the gas causes the mixture to be released as a fine spray (aerosol) or foam (aerogel) when a valve is opened.
atmosphere → atmosfera
1. Atmosphere is the column of air which is extending several hundred kilometers above the surface the Earth's surface. The density of this air decreases as you proceed up from the surface. The air in the atmosphere consists of 78 % nitrogen, 21 % oxygen, and 0.9 % argon. The remaining 0.1 % of the atmosphere consists of ozone, water vapor, carbon dioxide, methane, helium, and neon. The atmosphere is divided into different regions. The lowest two layers are the troposphere (the layer closest to the earth) and the stratosphere respectively. These two layers contain more than 99 % of the atmospheric molecules.
2. Standard atmosphere (atm) is an obsolete pressure and stress unit which should be discontinued. It is unit of pressure equal to the air pressure measured at mean sea level.
1 atm = 101 325 Pa
Technical atmosphere (at) is an obsolete MKpS pressure and sttress derived unit.
1 at = 98 066.5 Pa
1 atm = 1.033 227 453 at
autoxidation → autooksidacija
Autoxidation, autooxidation is a oxidation is caused by exposure to air. Rust is an example of autoxidation. Autoxidation makes ether taken from half-filled bottles very dangerous, because air oxidises ether to highly explosive organic peroxides.
Avogadro’s law → Avogadrov zakon
Avogadro’s law: Equal volumes of all gases contain equal numbers of molecules at the same pressure and temperature. The law, often called Avogadro’s hypothesis, is true only for ideal gases. It was proposed in 1811 by Italian chemist Amadeo Avogadro (1776-1856).
azeotrope → azeotropi
Azeotrope is a mixture of two liquids that boils at constant composition, i.e. the composition of the vapour is the same as that of the liquid. Azeotropes occur because of deviations in Raoult’s law leading to a maximum or minimum in the boiling point - composition diagram. The composition of an azeotrope depends on the pressure.
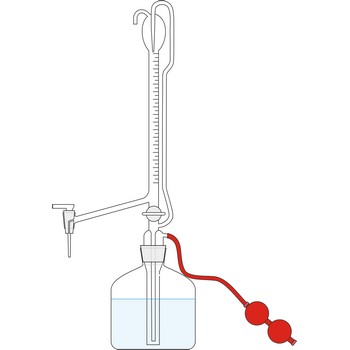
automatic burette → automatska bireta
Automatic burette is used for series of tests. It is connected with a bottle which contains the titration solution. The air is pumped into the bottle by a small rubber air pump, created the pressure in the bottle which the rises the solution to the top of burette. When the the burette is full, the valve is released, the pressure in the bottle falls and the burette automatically sets itself to zero. Work with automatic burettes is by far faster and the consumption of standard solution is smaller.
Citing this page:
Generalic, Eni. "Atmosferski tlak." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table