atmospheric pressure → atmosferski tlak
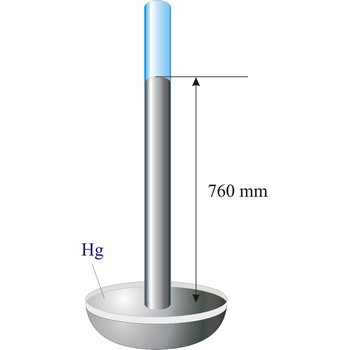
Atmospheric pressure is the pressure exerted by weight of the air above it at any point on the earth’s surface. At sea level the atmosphere will support a column of mercury about 760 mm high. This decreases with increasing altitude. The standard value for the atmospheric pressure at sea level in SI units is 101 325 Pa.
critical pressure → kritični tlak
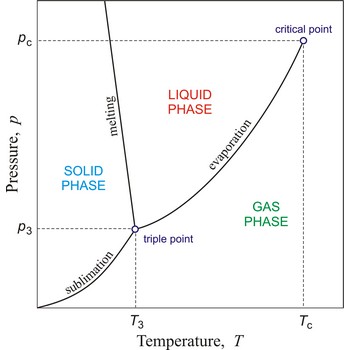
Critical pressure is the pressure of a fluid in its critical point; i.e. when it is at its critical temperature and critical volume.
partial pressure → parcijalni tlak
Partial pressure is a pressure that one component of gas mixture would have if it were alone in the same volume and at the same temperature as the mixture is in now.
vapour pressure → tlak pare
Vapour pressure is the pressure of a gas in equilibrium with a liquid (or, in some usage, a solid) at a specified temperature.
osmotic pressure → osmotski tlak
Osmotic pressure (Π) is the excess pressure necessary to maintain osmotic equilibrium between a solution and a pure solvent separated by a membrane permeable only to the solvent. In an ideal dilute solution
where cB is the amount-of-substance concentration of the solute, R is the molar gas constant, and T the temperature.
vapour pressure lowering → snižavanje tlaka pare
Vapour pressure is a colligative property of solutions. The vapour pressure of a solution is always lower than the vapour pressure of the pure solvent. Ratio of solution to pure solvent vapour pressures is approximately equal to the mole fraction of solvent in the solution.
barometer → barometar
Barometer is an instrument that measures atmospheric pressure. A mercury barometer is a closed tube filled with mercury inverted in a mercury reservoir. The height of the mercury column indicates atmospheric pressure (with 1 atm = 760 mm of mercury). An aneroid barometer consists of an evacuated container with a flexible wall. When atmospheric pressure changes, the wall flexes and moves a pointer which indicates the changing pressure on a scale.
boiling point → vrelište
Boiling point is the temperature at which the vapour pressure of a liquid is equal to the external pressure on the liquid. The standard boiling point is the temperature at which the vapour pressure of a liquid equals standard pressure (101 325 Pa).
Torricelli, Evangelista → Torricelli, Evangelista
Evangelista Torricelli (1852-1908) is Italian physicist and mathematician. He became the first scientist to create a sustained vacuum and to discover the principle of a barometer. He filled a tube three feet long, and hermetically closed at one end, with mercury and set it vertically with the open end in a basin of mercury, taking care that no air-bubbles should get into the tube. The column of mercury invariably fell to about twenty-eight inches, leaving an empty space above its level. He discovered that the variation of the height of the mercury from day to day was caused by changes in the atmospheric pressure. He also constructed a number of large objectives and small, short focus, simple microscopes.
Citing this page:
Generalic, Eni. "Atmosferski tlak." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table