iridium → iridij
Iridium was discovered by Smithson Tennant (England) in 1803. The origin of the name comes from the Latin word iris, meaning rainbow, because its salts are highly colored. It is heavy, brittle, white metal. Unreactive in air, water and acids. Attacked by fused NaOH. Metal ignites and burns readily. Iridium is found in gravel deposits with platinum. Used with osmium to tip gold pen points, to make crucible and special containers. Also to make alloys used for standard weights and measures and heat-resistant alloys. Also as hardening agent for platinum.
iron → željezo
Iron has been known since ancient times. The origin of the name comes from the Latin word ferrum meaning iron. It is malleable, ductile, silvery-white metal. Exposed surfaces form red-brown oxides. Forms very strong alloys (steel). Ferromagnetic. Metal dust flammable. Fourth most abundant element in the earth’s crust. Iron is obtained from iron ores. Pure metal produced in blast furnaces by layering limestone, coke and iron ore and forcing hot gasses into the bottom. This heats the coke red hot and the iron is reduced from its oxides and liquefied where it flows to the bottom. Iron is the most common metal in human society. More than 90 % of all metal refined in the world is iron. Used in steel and other alloys. It is the chief constituent of hemoglobin which carries oxygen in blood vessels. Its oxides are used in magnetic tapes and disks.
Joule-Thomson’s effect → Joule-Thomsonov efekt
Temperature of ideal gas will not be changed when it is repressed to a lower pressure, but when real gases are repressed to a lower pressure, a lower or higher temperature change appears under high pressures. The temperature change which appears at real gas expansion in a system into which energy is not brought is called Joule-Thomson’s effect. It was determined that when air is repressed by 1 bar, its temperature drops by 0.25 °C. That minute effect is completely irrelevant for most technical processes, but is also used in gas liquefying procedure.
Knudsen burette → Knudsenova bireta
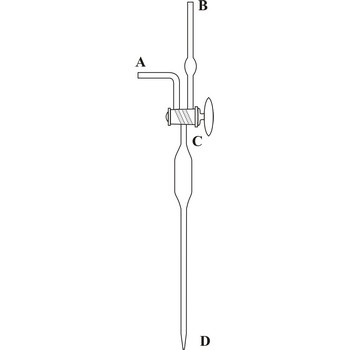
Knudsen's automatic bulb-burette, developed by the Danish physicist Martin Knudsen (1871-1949), is designed in a way that even routine field analysis in a boat laboratory would provide highly accurate measurements. The burette is filled with a mixture of silver nitrate from reservoir R, located above the burette, by opening the A valve. When the solution crosses the three-way C valve the A valve is closed preventing further solution flow in to the burette. Any extra solution is caught in the W bowl. Turn the C valve, which marks the zero on the scale, in order to allow atmospheric air to enter the burette. Since most open-ocean samples lie in a relatively small chlorinity range, the burette is designed so that much of its capacity is in the bulb (B). This allows the titration to be quick (by quickly releasing contents from the B area) and reduces the error that occurs from the slow drainage along the inner wall of the burette.
Each millimeter is divided in to twenty parts (double millimeter division of the Knudsen burette) which allows for highly accurate measurements (the scale is read up to a precision of 0.005 mL). From 0 to 16 the burette isn't divided, that usually starts from 16 and goes until 20.5 or 21.5. A single double millimeter on a Knudsen burette scale corresponds to one permille of chloride in the seawater sample. This burette can be used for titration of water from all of the oceans and seas, with the exemptions being areas with very low salinity (e.g. the Baltic Sea) and river estuaries which require the use of normal burettes.
Knudsen’s pipette → Knudsenova pipeta
Kudsen's automatic pipette, developed by the Danish physicist Martin Knudsen (1871-1949), allows quick and accurate transfer of a constant volume of liquid (sea water), usually around 15 mL. On the top of pipette is a double sided C vent that can establish flow between the body of the pipette and one of the branches (A or B), or isolate the body of the pipette from both of the branches. Sucking through the B branch the pipette is filled with liquid, it is closed with a twist of the C valve and the liquid is released by rotating the valve towards the A branch (so atmospheric air can enter the pipette). Emptying the pipette takes around 30 seconds. Before it's first use, the pipette must be calibrated with distilled water.
krypton → kripton
Krypton was discovered by Sir William Ramsay and Morris W. Travers (England) in 1898. The origin of the name comes from the Greek word kryptos meaning hidden. It is colourless, odourless rare noble gas. Reacts only with fluorine. Krypton is obtained from production of liquid air. Used in lighting products. Some is used as inert filler-gas in incandescent bulbs. Some is mixed with argon in fluorescent lamps. The most important use is in flashing stroboscopic lamps that outline airport runways.
lanthanum → lantan
Lanthanum was discovered by Carl Gustaf Mosander (Sweden) in 1839. The origin of the name comes from the Greek word lanthanein meaning to lie hidden. It is soft, silvery-white, malleable, ductile metal. Readily tarnishes in air. Reaction with water releases hydrogen gas. Metal ignites and burns readily. Reacts with oxidants. Lanthanum is found with rare earths in monazite and bastnasite. Monazite sand typical contains 25 % lanthanum. It is used in the electrodes of high-intensity, carbon-arc lights. Because it gives glass refractive properties, it is used in expensive camera lenses.
lead → olovo
Lead has been known since ancient times. The origin of the name comes from the Latin word plumbum meaning liquid silver. It is very soft, highly malleable and ductile, blue-white shiny metal. Tarnishes in moist air; stable in oxygen and water. Dissolves in nitric acid. Compounds toxic by inhalation or ingestion. Danger of cumulative effects. Lead is found most often in ores called galena or lead sulfide (PbS). Used in solder, shielding against radiation and in batteries.
lithium → litij
Lithium was discovered by Johan August Arfvedson (Sweden) in 1817. The origin of the name comes from the Greek word lithos meaning stone, apparently because it was discovered from a mineral source whereas the other two elements, sodium and potassium, were discovered from plant sources. It is soft silvery-white metal. Lightest of metals. Reacts slowly with water and oxygen. Flammable. Can ignite in air. Reacts with water to give off a flammable gas. Lithium is obtained by passing electric charge through melted lithium chloride and from the silicate mineral called spodumene [LiAl(Si2O6)]. Used in batteries. Also for certain kinds of glass and ceramics. Some is used in lubricants.
luminescence → luminiscencija
Luminescence (from Latin lumen, light) is the emission of electromagnetic radiation (UV, visible or IR) from atoms or molecules as a result of the transition of an electronically excited state to a lower energy state, usually the ground state. Luminescence can be divided into categories by duration (fluorescence or phosphorescence) or by the mechanism that creates the light (radioluminescence, electroluminescence, photoluminescence, thermoluminescence, triboluminescence, chemiluminescence, bioluminescence). The prefix identifies the energy source responsible for generating or releasing the light.
Phosphorescence is emission of light from a substance exposed to radiation and persisting as an afterglow after the source of excitation has been removed. Fluorescence, on the other hand, is an almost instantaneous effect, ending within about 10-8 second after excitation.
Citing this page:
Generalic, Eni. "X-zrake." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table