photochemical reaction → fotokemijska reakcija
Photochemical reactions are those reactions which are conducted under the influence of light that is under the influence of ultraviolet, visible and infrared part of the light spectrum. Some systems can be influenced only by radiation that is absorbed by that system. Photochemical reactions are for example photosynthesis, creation of photography, generation of phosgene, creation of hydrochloride etc.
photomultiplier → fotomultiplikator
Photomultiplier (photomultiplier tube or PMT) is a very versatile and sensitive detector of radiant energy in the ultraviolet, visible, and near infrared regions of the electromagnetic spectrum. A typical photomultiplier tube consists of a photoemissive cathode (photocathode) followed by focusing electrodes, an electron multiplier (dynode) and an electron collector (anode) in a vacuum tube.
ultraviolet light → ultraljubičasto svjetlo
Ultraviolet light (UV light or UV radiation) is an electromagnetic radiation with a wavelength longer than that of x-rays, but shorter than that of visible light. Ultraviolet light can break some chemical bonds and cause cell damage.
absorbance → apsorbancija
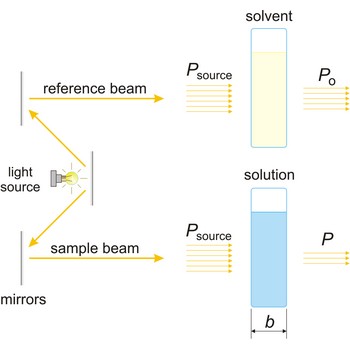
Absorbance (A) is a logarithm of the ratio of incident radiant power (Po) to transmitted radiant power (P) through a sample (excluding the effects on cell walls).
The absorption of light by a substance in a solution can be described mathematically by the Beer-Lambert law
where A is the absorbance at a given wavelength of light, ε is the molar absorbtivity or extinction coefficient (L mol-1 cm-1), unique to each molecule and varying with wavelength, b is the length of light path through the sample (cm), and c is the concentration of the compound in solution (mol L-1).
absorbed dose → apsorbirana doza
For any ionising radiation, absorbed dose (D) is the mean energy imparted to an element of irradiated matter divided by the mass of that element.
absorptance → apsorptancija
Absorptance (α) is the ratio of the radiant or luminous flux in a given spectral interval absorbed in a medium to that of the incident radiation. Also called absorption factor.
absorption coefficient → apsorpcijski koeficijent
Absorption coefficient (a) is the relative decrease in the intensity of a collimated beam of electromagnetic radiation, as a result of absorption by a medium, during traversal of an infinitesimal layer of the medium, divided by the length traversed.
alpha particle → alfa-čestica
Alpha particle is a helium nucleus emitted spontaneously from radioactive elements, both natural and manufactured. Its energy is in range 4-8 MeV and is dissipated in a very short path, i.e. a few centimetres of air or less than 0.005 mm of aluminium. As helium nucleus consists of two protons and two neutrons bound together as a stable entity the loss of an alpha particle involves a decrease in nucleon number of 4 and decrease of 2 in the atomic number, e.g.
A stream of alpha particles is known as an alpha ray or alpha-radiation.
atomic absorption spectroscopy → atomska apsorpcijska spektroskopija
Atomic absorption spectroscopy (AAS) An analytical technique in which a sample is vapourised and the nonexcited atoms absorb electromagnetic radiation at characteristic wavelengths.
autoxidation → autooksidacija
Autoxidation, autooxidation is a oxidation is caused by exposure to air. Rust is an example of autoxidation. Autoxidation makes ether taken from half-filled bottles very dangerous, because air oxidises ether to highly explosive organic peroxides.
Citing this page:
Generalic, Eni. "Vidljivo zračenje." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table