Joule-Thomson’s effect → Joule-Thomsonov efekt
Temperature of ideal gas will not be changed when it is repressed to a lower pressure, but when real gases are repressed to a lower pressure, a lower or higher temperature change appears under high pressures. The temperature change which appears at real gas expansion in a system into which energy is not brought is called Joule-Thomson’s effect. It was determined that when air is repressed by 1 bar, its temperature drops by 0.25 °C. That minute effect is completely irrelevant for most technical processes, but is also used in gas liquefying procedure.
kelvin → kelvin
Kelvin (K) is the SI base unit of thermodynamic temperature.
The kelvin, unit of thermodynamic temperature, is the fraction 1/273.16 of the thermodynamic temperature of the triple point of water. The unit was named after the British scientist Sir. W. Thompson, Lord Kelvin (1824-1907).kinetic energy → kinetička energija
Kinetic energy (Ek) is associated with the state of motion of a body. It is a scalar property and defined to be
Kinetic energy is most clearly exhibited in gases, in which molecules have much greater freedom of motion than in liquids and solids.
law of conservation of mass → zakon o očuvanju mase
Law of conservation of mass states that no detectable gain or loss in mass occurs in chemical reactions. The state of a substance may change in a chemical reaction, for example, from a solid to a gas, but its total mass will not change. Note that the energy released (exothermic) or adsorbed (endothermic) in a chemical reaction is a result of energy transfer between atoms and their environment.
logarithmic scale → logaritamska skala
Logarithmic scale is the one in which values of 1, 2, 3, 4, 5, in fact represents values of 1, 10, 100, 1 000, 10 000. Logarithmic scales are often used to simplify graphs and tables, where otherwise changes of data at the lower end of the scale would be difficult to distinguish (e.g. a graph axis which would normally have values from 1 - 1 000 000 is shown by values of 1 - 7). An example of a logarithmic scale is the pH scale.
luminescence → luminiscencija
Luminescence (from Latin lumen, light) is the emission of electromagnetic radiation (UV, visible or IR) from atoms or molecules as a result of the transition of an electronically excited state to a lower energy state, usually the ground state. Luminescence can be divided into categories by duration (fluorescence or phosphorescence) or by the mechanism that creates the light (radioluminescence, electroluminescence, photoluminescence, thermoluminescence, triboluminescence, chemiluminescence, bioluminescence). The prefix identifies the energy source responsible for generating or releasing the light.
Phosphorescence is emission of light from a substance exposed to radiation and persisting as an afterglow after the source of excitation has been removed. Fluorescence, on the other hand, is an almost instantaneous effect, ending within about 10-8 second after excitation.
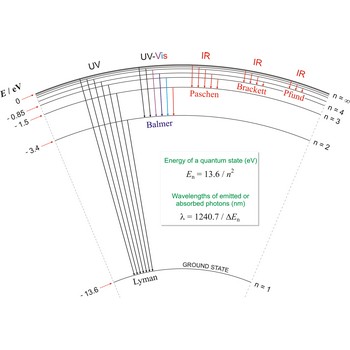
Lyman series → Lymanova serija
Lyman series is the series of lines in the spectrum of the hydrogen atom which corresponds to transitions between the ground state (principal quantum number n = 1) and successive excited states.
mass spectrometry → masena spectrometrija
Mass spectrometry is an analytical technique in which ions are separated according to the mass/charge (m/e) ratio and detected by a suitable detector.
In a mass spectrometer a sample is ionised and the positive ions produced are accelerated into a high-vacuum region containing electric and magnetic fields. These fields deflect and focus the ions onto a detector. A mass spectrum is thus obtained, consisting of a series of peaks of variable intensity to which m/e values can be assigned. Different molecules can be identified by their characteristic pattern of lines.
melting point → talište
Melting point is the temperature at which a solid becomes a liquid at normal atmospheric pressure.
A more specific definition of melting point (or freezing point) is the temperature at which the solid and liquid phases of a substance are in equilibrium at a specified pressure (normally taken to be atmospheric unless stated otherwise). A pure substance under standard condition of pressure has a single reproducible melting point. The terms melting point and freezing point are often used interchangeably, depending on whether the substance is being heated or cooled.
metal → metal
Metals are materials in which the highest occupied energy band (conduction band) is only partially filled with electrons.
Their physical properties generally include:
- They are good conductors of heat and electricity. The electrical conductivity of metals generally decreases with temperature.
- They are malleable and ductile in their solid state.
- They show metallic lustre.
- They are opaque.
- They have high density.
- They are solids (except mercury)
- They have a crystal structure in which each atom is surrounded by eight to twelve near neighbours
Their chemical properties generally are:
- They have one to four valence electrons.
- They have low ionisation potentials; they readily lose electrons.
- They are good reducing agents.
- They have hydroxides which are bases or amphoteric.
- They are electropositive.
Metallic characteristics of the elements decrease and non-metallic characteristics increase with the increase of valence electrons. Also metallic characteristics increase with the number of electron shells. Therefore, there is no sharp dividing line between the metals and non-metals.
Of the 114 elements now known, only 17 show primarily non-metallic characteristics, 7 others are metalloids, and 89 may be classed as metals.
Citing this page:
Generalic, Eni. "Promjena stanja." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table