elementary reaction → elementarna reakcija
Elementary reaction is a reaction that occurs in a single step. Equations for elementary reactions show the actual molecules, atoms, and ions that react on a molecular level.
first law of thermo-dynamics → prvi zakon termodinamike
First law of thermo-dynamics is: Energy can be neither created nor destroyed, but can cross from one shape to another.
glass → staklo
Glass is a brittle transparent solid with the molecular structure of a liquid. It is made by fusing together sand (SiO2), soda (Na2CO3), and lime (CaCO3) with small quantities other compounds. It is used for window panes and mirrors, for articles of table and culinary use, for lenses, and various articles of ornament.
intermediate → intermedijer
Intermediate is a molecular or ionic species that is formed (directly or indirectly) from the reactants and reacts further (directly or indirectly) to form the products of the reaction. It does not accumulate during the course of the reaction.
electrophoresis → elektroforeza
Electrophoresis is a technique for the analysis and separation of colloids, based on the movement of charged colloidal particles in an electric field. The migration is toward electrodes of charge opposite to that of the particles. The rate of migration of the particles depends on the field, the charge on the particles, and on other factors, such as the size and shape of the particles.
Electrophoresis is important in the study of proteins. The acidity of the solution can be used to control the direction in which a protein moves upon electrophoresis.
energy → energija
Energy (E, U) is the characteristic of a system that enables it to do work. Like work itself, it is measured in joules (J).
The internal energy of a body is the sum of the potential energy and the kinetic energy of its component atoms and molecules.
Potential energy is the energy stored in a body or system as a consequence of its position, shape, or state (this includes gravitation energy, electrical energy, nuclear energy, and chemical energy).
Kinetic energy is the energy of motion and is usually defined as the work that will be done by a body possessing the energy when it is brought to rest. For a body of mass m having a speed v, the kinetic energy is mv2/2. Kinetic energy is most clearly exhibited in gases, in which molecules have much greater freedom of motion than in liquids and solids.
In an isolated system energy can be transferred from one form to another but the total energy of the system remains constant.
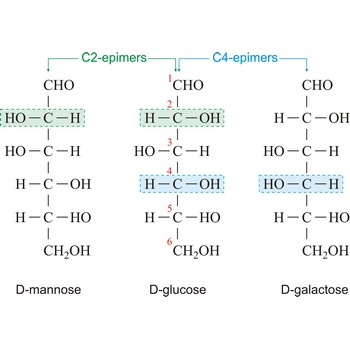
epimer → epimer
Epimers are diastereoisomers that have the opposite configuration at only one of two or more chiral centers present in the respective molecular entities. For example D-glucose and D-mannose, which differ only in the stereochemistry at C-2, are epimers, as are D-glucose and D-galactose (which differ at C-4).
Citing this page:
Generalic, Eni. "Linear molecular shape." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table