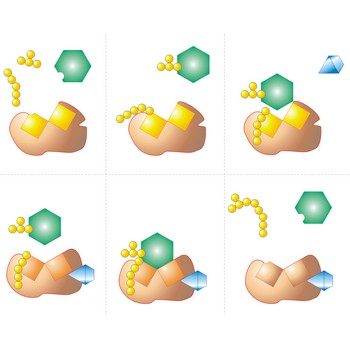
active site → aktivno mjesto
Active site is a pocket or crevice on an enzyme molecule that fits reactant molecules like a hand in a glove. The active site lowers the activation energy for reaction
AMU → AMU
AMU or atomic mass unit is a unit of mass used to express relative atomic masses. It is equal to 1/12 of the mass of an atom of the isotope carbon-12 and is equal to 1.66 033×10-27 kg. This unit superseded both the physical and a chemical mass unit based on oxygen-16 and is sometimes called the unified mass unit or the dalton.
anisotropy → anizotropija
Anisotropy is the property of molecules and materials to exhibit variations in physical properties along different molecular axes of the substance.
atomic clock → atomski sat
Atomic clock is an apparatus for standardizing time based on periodic phenomena within atoms or molecules (ammonia clock; caesium clock).
Avogadro, Amadeo → Avogadro, Amadeo
Amadeo Avogadro (1776-1856) is an Italian chemist and physicist that proposed a correct molecular explanation for Gay-Lussac’s law of combining volumes. His work provided a simple way to determine atomic weights and molecular weights of gases. He is published a theory about the movement of particles in gases that became known as Avogadro’s Law.
activity → aktivitet
Activity (a) is a thermodynamic function used in place of concentration in equilibrium constants for reactions involving nonideal gases and solutions. For the species i activity is defined as
where ai is the activity of the species i, ci is its molar concentration, and fi is a dimensionless quantity called the activity coefficient.
activity coefficient → koeficijent aktiviteta
Activity coefficient (γ or f) is a fractional number which, when multiplied by the molar concentration of a substance in solution, yields the chemical activity. This term gives an idea of how much interaction exists between molecules at higher concentration.
In solutions of very low ionic strength, when m is less than 0.01, the Debye-Hückel limiting law can be used to calculate approximate activity coefficients
where γi = activity coefficient of the species i, zi = charge on the species i and μ = ionic strength of the solution.
addition reactions → reakcije adicije
Addition reactions are normally occur with unsaturated compounds and involve the addition of one molecule (called the reactant) across the unsaturated bond (i.e. the double bond or the triple bond) of another molecule (called the substrate) to give a single product, formed by the combination of both reacting molecules.
For example, bromine adds across the double bond of ethene in an addition reaction to form dibromoethane.
alanine → alanin
Alanine is hydrophobic amino acids with aliphatic side chain. It is the second simplest amino acid, but used the most in proteins. The nonpolar hydrophobic amino acids tend to cluster together within proteins, stabilizing protein structure by means of hydrophobic interactions. Alanine is a nonessential amino acid, meaning it can be manufactured by the human body, and does not need to be obtained directly through the diet.
- Abbreviations: Ala, A
- IUPAC name: 2-aminopropanoic acid
- Molecular formula: C3H7NO2
- Molecular weight: 89.09 g/mol
Avogadro’s law → Avogadrov zakon
Avogadro’s law: Equal volumes of all gases contain equal numbers of molecules at the same pressure and temperature. The law, often called Avogadro’s hypothesis, is true only for ideal gases. It was proposed in 1811 by Italian chemist Amadeo Avogadro (1776-1856).
Citing this page:
Generalic, Eni. "Kiralne molekule." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table