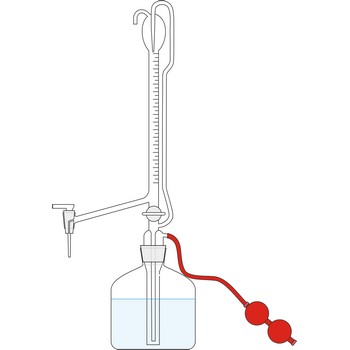
Schrotter apparatus for determination of CO2 → Schrotterova aparatura za određivanje CO2
Schrötter decomposition apparatus (Schrötter's alkalimeter) is used to determining the carbonate content in samples of limestone, gypsum, dolomite, or baking powder by loss of weight. The apparatus is named after the Austrian chemist Anton Schrötter von Kristelli (1802-1875), who devised it in 1871. The size of the filled apparatus (apparatus is 16 cm high) is such that it weights less than 75 g, and can be placed on the pan of an analytical balance.
Procedure: Weigh about 0.5 g of the powdered carbonate sample and introduce it into the decomposition flask C. Pour into the drying tube A 2-3 mL of concentrated sulphuric acid (H2SO4), and to the dropping funnel B add about 10-15 mL of hydrochloric acid (w(HCl) = 15 %). Weigh the whole apparatus. Open the upper taps of both parts and allow the hydrochloric acid from B to run slowly down on to the powdered sample. The evolved CO2 escapes through the strong sulphuric acid and is thus thoroughly dried. When further addition of acid produces no more evolution of CO2, warm the apparatus up to 80 °C so as to expel the CO2 from the solution. Connect the upper tap of the drying tube A to a water pump and draw a slow current of air through the apparatus until completely cool. Open the upper taps for a moment to equalize the internal and external pressure and weight the apparatus again. The weight loss is equal to the weight of carbon dioxide liberated from the carbonates.
carbon dating → određivanje starosti radioaktivnim ugljikom
Carbon dating is used to the time passed since a living organism died. It is based on measuring the quantity of isotope of carbon-14 that is contained in all living organisms.
activity coefficient → koeficijent aktiviteta
Activity coefficient (γ or f) is a fractional number which, when multiplied by the molar concentration of a substance in solution, yields the chemical activity. This term gives an idea of how much interaction exists between molecules at higher concentration.
In solutions of very low ionic strength, when m is less than 0.01, the Debye-Hückel limiting law can be used to calculate approximate activity coefficients
where γi = activity coefficient of the species i, zi = charge on the species i and μ = ionic strength of the solution.
amperometry → amperometrija
Amperometry is determining the concentration of a material in a sample by measuring electric current passing through a cell containing the solution.
automatic burette → automatska bireta
Automatic burette is used for series of tests. It is connected with a bottle which contains the titration solution. The air is pumped into the bottle by a small rubber air pump, created the pressure in the bottle which the rises the solution to the top of burette. When the the burette is full, the valve is released, the pressure in the bottle falls and the burette automatically sets itself to zero. Work with automatic burettes is by far faster and the consumption of standard solution is smaller.
Avogadro, Amadeo → Avogadro, Amadeo
Amadeo Avogadro (1776-1856) is an Italian chemist and physicist that proposed a correct molecular explanation for Gay-Lussac’s law of combining volumes. His work provided a simple way to determine atomic weights and molecular weights of gases. He is published a theory about the movement of particles in gases that became known as Avogadro’s Law.
blank determination → slijepa proba
Blank determination is a procedure of determining which follows all steps of analysis but in the absence of a sample. It is used for detection and compensation of systematic analysis mistakes.
chemical equation equalization → izjednačavanje kemijske jednadžbe
Chemical equation equalization is determining values of stechiometric coefficients of reactants and products in a chemical equation in a way that the number of atoms of each element is equal before and after the reaction.
chemical technology → kemijska tehnologija
Chemical technology is a branch of applied chemistry that concerns technical methods and devices in order to manufacture a chemical product.
Citing this page:
Generalic, Eni. "Schrotterova aparatura za određivanje CO2." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table