Nessler’s reagent → Nesslerov reagens
Nessler’s reagent is a solution of mercury(II) iodide (HgI2) in potassium iodide and potassium hydroxide named after the German chemist Julius Nessler (1827-1905). It is used in testing for ammonia, with which it forms a brown coloration or precipitate.
Grignard reagent → Grignardov reagens
Grignard reagents are organomagnesium halides, RMgX, having a carbon- magnesium bond (or their equilibrium mixtures in solution with R2Mg + MgX2).
masking reagent → maskirajući reagens
Masking reagent reacts with sample components which can increase the analysis results.
Zimmermann-Reinhardt’s reagent → Zimmermann-Reinhardtov reagens
Zimmermann-Reinhardt’s reagent is a mixture of manganese(II) sulphate, sulphuric acid and phosphorus acid. It is used for preventing oxidation of chloride ion while titrating iron(II) ion with permanganate solution.
analytical balance → analitička vaga
Analytical balances are instruments used for precise determining mass of matter. Analytical balances are sensitive and expensive instruments, and upon their accuracy and precision the accuracy of analysis result depends. The most widely used type of analytical balances are balances with a capacity of 100 g and a sensitivity of 0.1 mg. Not one quantitative chemical analysis is possible without usage of balances, because, regardless of which analytical method is being used, there is always a need for weighing a sample for analysis and the necessary quantity of reagents for solution preparation.
The working part of the balance is enclosed in a glass-fitted case. The baseplate is usually of black glass or black slate. The beam has agate knife-edges at its extremes, supporting stirrups from which balance pans are suspended. Another agate or steel knife-edge is fixed exactly in the middle of the beam on its bottom side. This knife-edge faces downwards and supports the beam. When not in use and during loading or unloading of the pans, the balance should be arrested.
The principle of operation of a modern laboratory balance bears some resemblance to its predecessor - the equal arm balance. The older instrument opposed the torque exerted by an unknown mass on one side of a pivot to that of an adjustable known weight on the other side. When the pointer returned to the center position, the torques must be equal, and the weight was determined by the position of the moving weights.
Modern electronic laboratory balances work on the principle of magnetic force restoration. In this system, the force exerted by the object being weighed is lifted by an electromagnet. A detector measures the current required to oppose the downward motion of the weight in the magnetic field.
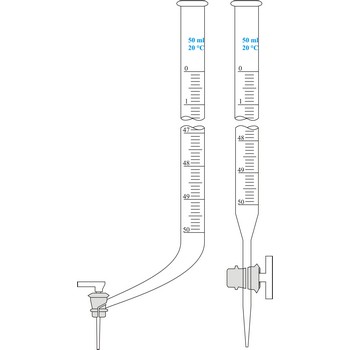
burette → bireta
Burette is a graded glass pipe which on its lower side has a glass faucet by which it can drop a precise quantity of liquid. Inner diameter of a burette must be equal in its whole length, because the accuracy of volume measurement depends upon that. Burettes are primarily used in volumetric analysis for titration with standard solution reagent. Most often Schellbach’s burette is used, graded on 50 mL with division of scale on 0.1 mL. Every burette is calibrated on discharge. For serial determining automatic burettes are used.
carboxylates → karboksilati
Carboxylates is a common name for all salts that carboxylic acids yield by reacting with hydroxides, carbonates, bicarbonates and other alkaline reagents.
limiting reactant → mjerodavni reaktant
Limiting reactant is a reactant in a chemical reaction that limits the amount of product that can be formed. The reaction will stop when the entire limiting reagent is consumed. These other reactants are present in excess.
desiccator → eksikator
Desiccator is a glass container with dry atmosphere due to the presence of some dehydrating agent. It is used for protecting the samples, reagents or precipitates from humidity. As dehydrating agent usually waterless calcium chloride (CaCl2) is used.
Citing this page:
Generalic, Eni. "Nessler%E2%80%99s reagent." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table