crystal system → kristalni sustav
Crystal system is a method of classifying crystalline substances on the basis of their unit cell. There are seven unique crystal systems. The simplest and most symmetric, the cubic (or isometric) system, has the symmetry of a cube. The other six systems, in order of decreasing symmetry, are hexagonal, tetragonal, rhombohedral (also known as trigonal), orthorhombic, monoclinic and triclinic.
|
Crystal system
|
Unit-cell
|
Conditions on unit-cell edges and angles |
|
cubic |
 |
a=b=c α=β=γ=90° |
|
hexagonal |
 |
a≠c α=γ=90° β=120° |
|
tetragonal |
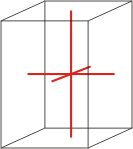 |
a=b≠c α=β=γ=90° |
|
rhombohedral |
 |
a=b=c α=β=γ≠90° |
|
orthorhombic |
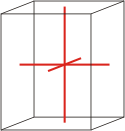 |
a≠b≠c α=β=γ=90° |
|
monoclinic |
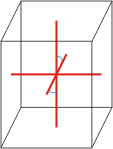 |
a≠b≠c α=γ=90°≠β |
|
triclinic |
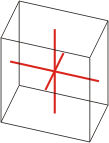 |
a≠b≠c α≠β≠γ≠90° |
sand filtration → pješčana filtracija
Sand filtration is a frequently used and very robust method of removing suspended solids from water. The filtration medium consists of a multiple layer of sand with a variety in size and specific gravity. Sand filters can be supplied in different sizes and materials, both hand operated and fully automatically.
toxic chemical → bojni otrov
Toxic chemical means any chemical which through its chemical action on life processes can cause death, temporary incapacitation, or permanent harm to humans or animals. This includes all such chemicals, regardless of their origin or of their method of production, and regardless of whether they are produced in facilities, in munitions or elsewhere.
ether → eter
Ethers are organic compounds with a formula R-O-R, where R is not equal to H. They may be derived from alcohols by elimination of water, but the major method is catalytic hydration of olefins. They are volatile highly flammable compounds; when containing peroxides they can detonate on heating. The term ether is often used synonymously with diethyl ether.
extraction → ekstrakcija
Extraction is the separation of a component from its mixture by selective solubility. When a solution of one substance in one solvent is brought in with another solvent dissolved substance will distribute between the two solutants because of different solubility. Extraction is an efficient and fast method used for separating and concentrating matters. Extraction is best done several times in a succession, with smaller amount of solvent in it the matter is better dissolved. For example, caffeine can be separated from coffee beans by washing the beans with supercritical fluid carbon dioxide; the caffeine dissolves in the carbon dioxide, but flavour compounds do not. Vanillin can be extracted from vanilla beans by shaking the beans with an organic solvent, like ethanol.
Fischer-Tropsch process → Fischer-Tropschov postupak
Fischer-Tropsch process is an industrial method of making hydrocarbon fuels from carbon monoxide and hydrogen. The process was introduced in 1933. and used by Germany in World War II. to produce motor fuel. Hydrogen and carbon monoxide are mixed in the ratio 2:1 (water gas was used with added hydrogen) and passed at 200 °C over a nickel or cobalt catalyst. The resulting hydrocarbon mixture can be separated into a higher-boiling fraction for Diesel engines and a lower-boiling petrol fraction. The petrol fraction contains a high proportion of straight-chain hydrocarbons and has to be reformed for use in motor fuel. Alcohols, aldehydes, and ketones are also present. The process is also used in the manufacture of SNG from coal. It is named after the German chemist Franz Fischer (1852-1932) and the Czech Hans Tropsch (1839-1935).
fluorine → fluor
Fluorine was discovered by Henri Moissan (France) in 1886. The origin of the name comes from the Latin word fluere meaning to flow. It is pale yellow to greenish gas, with an irritating pungent odour. Extremely reactive, flammable gas. Reacts violently with many materials. Toxic by inhalation or ingestion. Does not occur uncombined in nature. Fluorine is found in the minerals fluorite (CaF2) and cryolite (Na3AlF6). Electrolysis of hydrofluoric acid (HF) or potassium acid fluoride (KHF2) is the only practical method of commercial production. Used in refrigerants and other fluorocarbons. Also in toothpaste as sodium fluoride (NaF).
Frasch proces → Fraschov postupak
Frasch proces is a method of obtaining sulphur from underground deposits using a tube consisting of three concentric pipes. Superheated steam is passed down the outer pipe to melt the sulphur, which is forced up through the middle pipe by compressed air fed through the inner tube. The steam in the outer casing keeps the sulphur molten in the pipe. It was named after the German-born American chemist Herman Frasch (1851-1914).
gas washing bottle → boca ispiralica
Gas washing bottle or Drechsel bottle provide an inexpensive but effective method for washing or drying gases. The gas enters the bottle through the top of the central vertical tube, the lower end of which is below the surface of the washing medium. To maximize surface area contact of the gas to the liquid, a gas stream is slowly blown into the vessel through the fritted glass tip so that it breaks up the gas into many tiny bubbles. After bubbling through the medium, the gas rises to the top and exits through the side tube. It is named after the German chemist Edmund Drechsel (1843-1897).
Goldschmidt process → Goldschmidtov postupak
Goldschmidt process (thermite process) is a method of extracting metals by reducing the oxide with aluminium powder. Practically all the metallic oxides are reducible by this method, the chief exception being the oxide of magnesium. The thermite process was developed by the German chemist Hans Goldschmidt (1861-1923) in 1893.
Goldschmidt was originally interested in producing very pure metals, but he soon realized the value in welding, a process known as Thermit welding.
Citing this page:
Generalic, Eni. "Kjeldhal’s method." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table



