diffusion current → difuzijska struja
Diffusion current (id) is a current which is limited by the speed of particle diffusion in an electrolyte solution.
Beer’s law → Beerov zakon
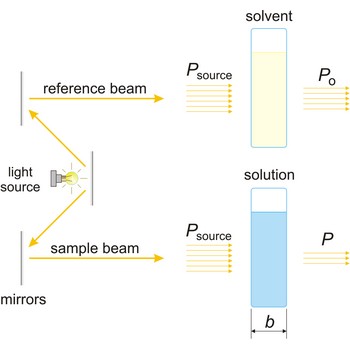
Beer’s law (or Beer-Lambert law) is the functional relationship between the quantity measured in an absorption method (A) and the quantity sought, the analyte concentration (c). As a consequence of interactions between the photons and absorbing particles, the power of the beam is attenuated from Po to P. Beer’s law can be written
where A is the absorbance at a given wavelength of light, ε is the molar absorbtivity or extinction coefficient (L mol-1 cm-1), unique to each molecule and varying with wavelength, b is the length of light path through the sample (cm), and c is the concentration of the compound in solution (mol L-1).
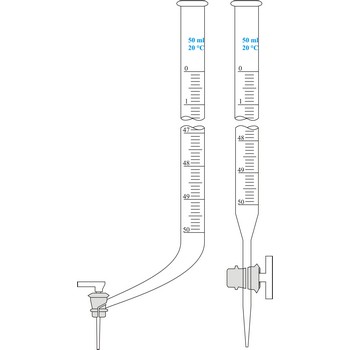
burette → bireta
Burette is a graded glass pipe which on its lower side has a glass faucet by which it can drop a precise quantity of liquid. Inner diameter of a burette must be equal in its whole length, because the accuracy of volume measurement depends upon that. Burettes are primarily used in volumetric analysis for titration with standard solution reagent. Most often Schellbach’s burette is used, graded on 50 mL with division of scale on 0.1 mL. Every burette is calibrated on discharge. For serial determining automatic burettes are used.
digestion → digeriranje
Digestion or precipitate ageing happens when freshly formed precipitate are left, usually at a higher temperature, in a solution from which it is precipitated. It results in cleaner and bigger particles.
dissolved substance → otopljena tvar
Dissolved substance is a solid, liquid or gas matter dissolved in a solvent. Depending upon the particle size of dissolved substance, solutions differ in properties and can be divided into real solutions (diameter of particles is smaller than 1 nm), colloid solutions (diameter of particles is from 1 nm to 200 nm) and suspensions (diameter of particles is greater than 200 nm).
Donnan potential → Donnanov potencijal
Donnan potential is the electrical potential difference between two solutions separated by an ion-exchange membrane in the absence of any current flowing through the membrane
elution → eluacija
Elution is the process of removing an adsorbed material (adsorbate) from an adsorbent with a liquid (eluent). The solution consisting of the adsorbate dissolved in the eluent is the eluate. Elution is the process used to wash components of a mixture through a chromatography column.
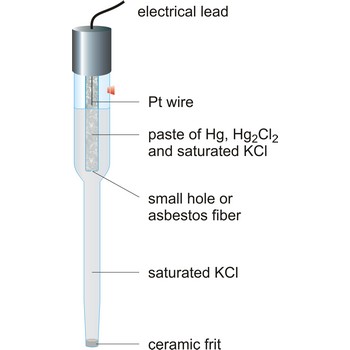
calomel electrode → kalomel elektroda
Calomel electrode is a type of half cell in which the electrode is mercury coated with calomel (Hg2Cl2) and the electrolyte is a solution of potassium chloride and saturated calomel. In the calomel half cell the overall reaction is
Table: Dependence of potential of calomel electrode upon temperature and concentration of KCl according to standard hydrogen electrode
| Potential vs. SHE / V | |||
|---|---|---|---|
| t / °C | 0.1 mol dm-3 | 3.5 mol dm-3 | sat. solution |
| 15 | 0.3362 | 0.254 | 0.2511 |
| 20 | 0.3359 | 0.252 | 0.2479 |
| 25 | 0.3356 | 0.250 | 0.2444 |
| 30 | 0.3351 | 0.248 | 0.2411 |
| 35 | 0.3344 | 0.246 | 0.2376 |
colloid ion → koloidni ion
Colloid ions emerge when colloid particles adsorb certain type of ion from solution and thus become charged with the same charge. The charge can also originate form a chemical reaction of colloid particle’s surface. Colloid ions formed by absorption of silver chloride particle can be show as follows:
Adsorbed layer is monomolecular (one molecule thick) and which type of ion will be formed depends upon which ions are present in a greater number in the solution in. Because of this colloid particles are charged with the same charge, mutual repelling occurs, and the colloid solution becomes stable. Colloid charge can be determined by electrophoresis.
complexometry → kompleksometrija
Complexometry is a volumetric analytic method which is based on titration of metal ion solutions with a substance that, combined with metal ions yields complex compounds, e.g. EDTA. The stoichiometric ratio of EDTA-metal in complexometric analyses is always 1:1, whatever the valency of the metal
Citing this page:
Generalic, Eni. "Binary solution." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table