Bunsen’s cell → Bunsenov članak
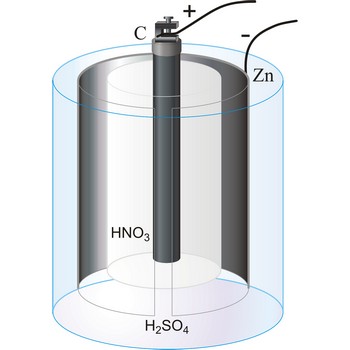
Bunsen’s cell is a primary cell devised by Robert W. Bunsen consisting of a zinc cathode immersed in dilute sulphuric acid and carbon anode immersed in concentrated nitric acid. The electrolytes are separated by a porous pot. The cell gives an e.m.f. of about 1.9 V.
burette → bireta
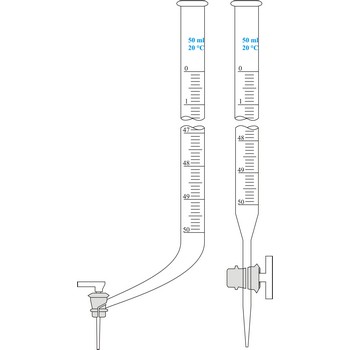
Burette is a graded glass pipe which on its lower side has a glass faucet by which it can drop a precise quantity of liquid. Inner diameter of a burette must be equal in its whole length, because the accuracy of volume measurement depends upon that. Burettes are primarily used in volumetric analysis for titration with standard solution reagent. Most often Schellbach’s burette is used, graded on 50 mL with division of scale on 0.1 mL. Every burette is calibrated on discharge. For serial determining automatic burettes are used.
cycloalkanes → cikloalkani
Cycloalkanes are cyclic saturated hydrocarbons containing a ring of carbon atoms joined by single bonds. They have the general formula CnH2n, for example cyclohexane, C6H12. In general, they behave like the alkanes but are rather less reactive.
diffusion current → difuzijska struja
Diffusion current (id) is a current which is limited by the speed of particle diffusion in an electrolyte solution.
digestion → digeriranje
Digestion or precipitate ageing happens when freshly formed precipitate are left, usually at a higher temperature, in a solution from which it is precipitated. It results in cleaner and bigger particles.
cadmium → kadmij
Cadmium was discovered by Friedrich Strohmeyer (Germany) in 1817. The origin of the name comes from the Latin word cadmia meaning calamine (zinc carbonate, ZnCO3), or from the Greek word kadmeia with the same meaning. It is soft, malleable, blue-white metal. Tarnishes in air, soluble in acids, insoluble in alkalis. Boiling cadmium gives off a weird, yellow-colored vapour that is poisonous. Cadmium can cause a variety of health problems, including kidney failure and high blood pressure. Cadmium is obtained as a by product of zinc refining. The mayor use of cadmium is in electroplating of steel to protect it from corrosion. Also used to make nickel-cadmium batteries. The ability of cadmium to adsorb neutrons has made it of great importance in the design of nuclear reactors. Its compounds are found in paint pigments and a wide variety of intense colours.
calomel electrode → kalomel elektroda
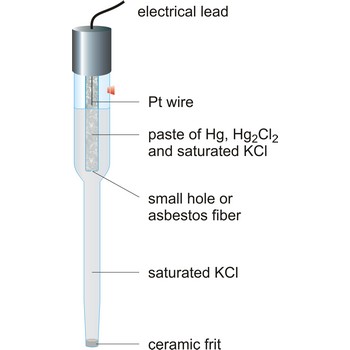
Calomel electrode is a type of half cell in which the electrode is mercury coated with calomel (Hg2Cl2) and the electrolyte is a solution of potassium chloride and saturated calomel. In the calomel half cell the overall reaction is
Table: Dependence of potential of calomel electrode upon temperature and concentration of KCl according to standard hydrogen electrode
| Potential vs. SHE / V | |||
|---|---|---|---|
| t / °C | 0.1 mol dm-3 | 3.5 mol dm-3 | sat. solution |
| 15 | 0.3362 | 0.254 | 0.2511 |
| 20 | 0.3359 | 0.252 | 0.2479 |
| 25 | 0.3356 | 0.250 | 0.2444 |
| 30 | 0.3351 | 0.248 | 0.2411 |
| 35 | 0.3344 | 0.246 | 0.2376 |
dilution → razrjeđivanje
Dilution is the action of diluting or reducing the strength or concentration of a liquid, usually by the addition of water.
dissolved substance → otopljena tvar
Dissolved substance is a solid, liquid or gas matter dissolved in a solvent. Depending upon the particle size of dissolved substance, solutions differ in properties and can be divided into real solutions (diameter of particles is smaller than 1 nm), colloid solutions (diameter of particles is from 1 nm to 200 nm) and suspensions (diameter of particles is greater than 200 nm).
Donnan potential → Donnanov potencijal
Donnan potential is the electrical potential difference between two solutions separated by an ion-exchange membrane in the absence of any current flowing through the membrane
Citing this page:
Generalic, Eni. "Zasićena otopina." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table