henry → henri
Henry (H) is the SI derived unit of inductance equal to the inductance of a closed circuit in which an e.m.f. of one volt is produced when the electric current in the circuit varies uniformly at a rate of one ampere per second (H = V·s/A). The unit was named after the American physicst Joseph Henry (1797-1878).
concentration → koncentracija
1. Group of four quantities characterizing the composition of a mixture with respect to the volume of the mixture (mass, amount, volume and number concentration).
2. Short form for amount (of substance) concentration (substance concentration in clinical chemistry).
critical pressure → kritični tlak
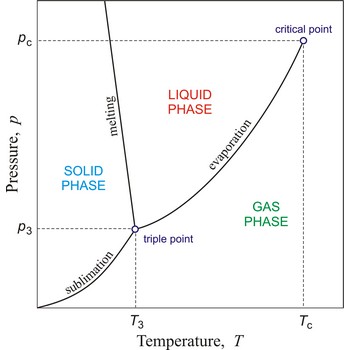
Critical pressure is the pressure of a fluid in its critical point; i.e. when it is at its critical temperature and critical volume.
cryogenic fluid → kriogenski fluid
Cryogenic fluids are used for accessing low temperatures, usually below -150 °C. Cryogenic temperatures are achieved by the rapid evaporation of volatile liquids. The most common laboratory cryogenic fluids are liquid nitrogen (-196 °C). Nitrogen gas is colorless and odorless. The cloud formed when pouring liquid nitrogen is condensed water vapour from the air, not nitrogen gas.
density → gustoća
In the most common usage, density (ρ) is mass density or mass per unit volume. In Si units it is measured in kg m-3. More commonly, densities are given in kg dm-3.
More generally, it is the amount of some quantity (mass, charge, energy, etc.) divided by a length, area, or volume.
Relative density is the ratio of the density of a substance to the density of some reference substance. For liquids or solids, it is the ratio of the density (usually at 20 °C) to the density of water at 4 °C. This quantity was formerly called specific gravity.
molar quantity → molarna veličina
Molar quantity is often convenient to express an extensive quantity (e.g., volume, enthalpy, heat capacity, etc.) as the actual value divided by the amount of substance (number of moles). The resulting quantity is called molar volume, molar enthalpy, etc.
ohm → om
Ohm (Ω) is the SI derived unit of electric resistance. The ohm is the electric resistance between two points of a conductor when a constant difference of potential of one volt, applied between these two points, produces in this conductor a current of one ampere, this conductor not being the source of electromotive force (Ω = V/A). The unit was named after the German physicist Georg Simon Ohm (1789-1854).
diamond → dijamant
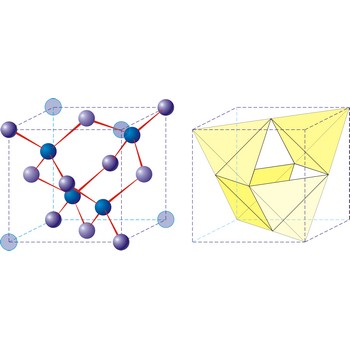
Diamond is the hardest known mineral (with a hardness of 10 on Mohs’ scale). It is an allotropic form of pure carbon that has crystallised in the cubic system, usually as octahedral or cubes, under great pressure. Diamond crystals my be colourless and transparent or yellow, brown or black. They are highly prized as gemstones, but also have extensive uses in industry, mainly for cutting and grinding tools. Diamonds occur in ancient volcanic pipes of kimberlite, or in river deposits that have been derived from weathered kimberlite. Industrial diamonds are being increasingly synthetically produced.
Citing this page:
Generalic, Eni. "Volt." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table