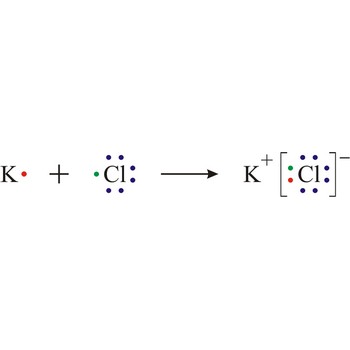wavefunction → valna funkcija
Wavefunction (Ψ) is a mathematical function that gives the amplitude of a wave as a function of position (and sometimes as a function of time and/or electron spin). Wavefunctions are used in chemistry to represent the behaviour of electrons bound in atoms or molecules.
indium → indij
Indium was discovered by Ferdinand Reich and Hieronymus Theodor Richter (Germany) in 1863. Named after the indicum (colour indigo), the colour it shows in a spectroscope. It is rare, very soft, silver-white metal. Stable in air and water. Dissolves in acids. Metal can ignite and burn. Indium is found in certain zinc ores. Used to coat high speed bearings and as an alloy that lowers the melting point of other metals. Relatively small amounts are used in dental items and in electronic semiconductors.
infrared radiation → infracrveno zračenje
Infrared radiation is an electromagnetic radiation within the area from 1.0 μm to 300 μm, and is responsible for the transmission of radiant heat.
wavelength → valna duljina
Wavelength is the distance between two neighbouring peaks of the electromagnetic wave.
weak base → slaba baza
Weak base is a base that only partially dissociates into ions in solution. Weak bases are weak electrolytes. Ammonia is an example of a weak base
zeta potential → zeta potencijal
Zeta potential (ζ) is the potential across the interface of all solids and liquids. Specifically, the potential across the diffuse layer of ions surrounding a charged colloidal particle, which is largely responsible for colloidal stability. Also called electrokinetic potential.
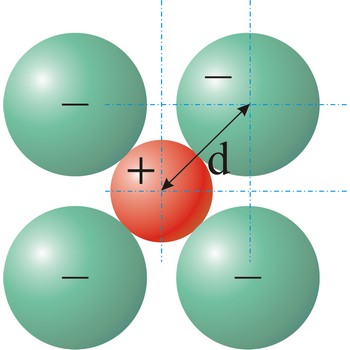
ionic bond → ionska veza
Ionic bond is a strong force of attraction holding atoms together in a molecule or crystal. Typically chemical bonds have energies of about 100 kJ mol-1. Ionic bond is a bond at which one of the participants, during the procedure of bonding, gives away its unpaired electrons to another atom so that both can achieve electron arrangement of the closest noble gas. In order to form an ionic bond one of the atoms must cross to the positively charged ion by losing certain number of electrons and the other atom must receive those electrons and cross to the negatively charged ion.
ionic radius → ionski radijus
Ionic radius is the radius of anions and cations in crystalline ionic compounds, as determined by consistently partitioning the center-to-center distance of ions in those compounds. In general, negative ions have larger ionic radii than positive ions.
isoelectric point → izoelektrična točka
Isoelectric point (pI or IEP) is the pH of a solution or dispersion at which the net charge on the molecules or colloidal particles is zero. In electrophoresis there is no motion of the particles in an electric field at the isoelectric point. The net charge (the algebraic sum of all the charged groups present) of any amino acid, peptide or protein, will depend upon the pH of the surrounding aqueous environment. For example, alanine can have a charge of +1, 0, or -1, depending on the pH of the solution in which it is dissolved.
Kohlrausch’s law → Kohlrauschov zakon
Kohlrausch’s law states that the equivalent conductivity of an electrolyte at infinite dilution is equal to the sum of the conductances of the anions and cations. If a salt is dissolved in water, the conductivity of the solution is the sum of the conductances of the anions and cations. The law, which depends on the independent migration of ions, was deduced experimentally by the German chemist Friedrich Kohlrausch (1840-1910).
Citing this page:
Generalic, Eni. "Valentni elektron." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table